In each part, an orbital diagram for an atom is given. Identify the element and whether this
Question:
In each part, an orbital diagram for an atom is given. Identify the element and whether this is the ground state of the atom. For any excited states, show the orbital diagram for the ground state.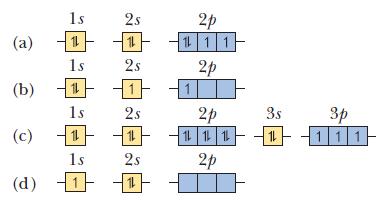
Transcribed Image Text:
(a) (b) (c) (d) 1s 11 15 1s 11 15 1 2s 11 25 2s 2s 1 2p 1 1 1 2p 2p 是一 1 1 1 20 3s 12 p 111
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
a O ground stat...View the full answer

Answered By

Carly Cimino
As a tutor, my focus is to help communicate and break down difficult concepts in a way that allows students greater accessibility and comprehension to their course material. I love helping others develop a sense of personal confidence and curiosity, and I'm looking forward to the chance to interact and work with you professionally and better your academic grades.
4.30+
12+ Reviews
21+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
In each part, an orbital diagram for an atom is given. Identify the element and whether this is the ground state of the atom. For any excited states, show the orbital diagram for the ground state. 1s...
-
Given the valence electron orbital level diagram and the description, identify the element or ion. a. A ground state atom b. An atom in an excited state (assume two electrons occupy the 1s orbital)...
-
Write an orbital diagram for the ground state of the phosphorus atom (Z = 15). Write all orbitals.
-
Computer Technologies provides maintenance service for computers and office equipment for companies throughout the Northeast. The sales manager is elated because she closed a $300,000 three-year...
-
A square plate is produced by welding together four smaller square plates, each of side a as shown in figure. Plate 1 weighs 40 N; plate 2, 60 N; plate 3, 30 N; and plate 4, 50 N. Find the center of...
-
Harry Samson opened a medical practice specializing in surgery. During the first month of operation (March), the business, titled Harry Samson, Professional Corporation (P.C.), experienced the...
-
Repeat Problem 9.4 without centering the regressors and compare the results. Which approach do you think is better? Data From Problem 9.4 Find the condition indices and the variance decomposition...
-
Tiffany Goren started her own consulting firm, Goren Consulting, on May 1, 2010. The trial balance at May 31 is as shown below. In addition to those accounts listed on the trial balance, the chart of...
-
Boyd Corporation issued $1,500,000 of 9% nonconvertible bonds at 107, due in 10 years. Each $1,000 bond was issued with 45 detachable stock warrants, each of which entitled the holder to purchase,...
-
The number 6 has four divisorsnamely, 1, 2, 3, and 6. List all numbers less than 20 that have exactly four divisors.
-
Give the maximum number of electrons that may occupy the following shells or subshells. (a) The 3p subshell (b) The 4d subshell (c) The fourth principal shell (d) The third principal shell
-
The speed of sound waves in air is 344 m/s, and the frequency of middle C is 512 Hz. What is the wavelength (in m) of this sound wave?
-
True or false? The O 2 and F anions have identical electron configurations. Justify your answer with Bohr diagrams and electron configuration notation.
-
Can an organization function without control? Comment.
-
Develop a different organizational structure for your company or college. What would be the advantages of the newly suggested structure over the existing organizational structure?
-
Is the grid analysis method a technique for doing analysis for each of the options based on the factors used to arrive at a decision? True False
-
How will you review your subordinates performance regularly and provide them with feedback about their work?
-
Which of these is also called T-group training? Role enactment In-basket training Sensitivity training Simulation training
-
The Steelcase Workplace Index studied the types of workrelated activities that Americans did while on vacation in the summer. Among other things, 40% read work-related material. Thirty-four percent...
-
A fast-food restaurant averages 150 customers per hour. The average processing time per customer is 90 seconds. a. Determine how many cash registers the restaurant should have if it wishes to...
-
How many rotational degrees of freedom are there for linear and nonlinear molecules?
-
Assuming 19 F 2 and 35 Cl 2 have the same bond length, which molecule do you expect to have the largest rotational constant?
-
Consider the rotational partition function for a polyatomic molecule. Can you describe the origin of each term in the partition function, and why the partition function involves a product of terms?
-
Can you set up a trading strategy using long and short positions of the default-free floating rate bond and the defaultable floating rate bond that replicates the cash flow pattern of the CDS?
-
The Capital Pricing Asset Model (CAPM) is frequently used to estimate a firm's cost of equity. a) use own words, explain how CAPM works. (3 marks) b) How do we derive a firm's equity beta? What...
-
You've entered the second-round interview of the bank. The manager asked you to analyze AMC Entertainment's financial ratios in 2019 and give comments . Your analysis must include 5 or more ratios...

Study smarter with the SolutionInn App


