Name the following compounds.
Question:
Name the following compounds.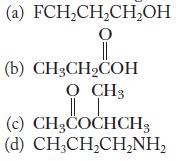
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
a 3fluoro1prop...View the full answer

Answered By

Umber Talat
I am providing full time mentoring and tutoring services in Business Finance, Contemporary issue in Global Economy, Quantitative Techniques, Principles of Marketing, strategic marketing, International Marketing, Organizational Behavior (OB), Consumer Behavior, Sales Force Management, Strategic Brand Management, Services Marketing, Integrated Marketing Communication (IMC), Principles of Management, General Management, Strategic Management, Small and Medium Enterprise Management, Innovation Management, Change Management, Knowledge Management, Strategic Planning, Operations Management, Supply Chain Management, Logistics Management, Inventory management, Total Quality Management (TQM), Productions Management, Project Management, Production Planning, Human Resource Management (HRM), Human Resource Development, Strategic HRM, Organizational Planning, Performance and Compensation Management, Recruitment and Selection, Organizational Development, Global Issues in Human Resource Management, Retail Marketing, Entrepreneurship, Entrepreneurial Marketing, International Business, Research Methods in Business, Business Communication, Business Ethics.
4.70+
158+ Reviews
236+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Name the following compounds by IUPAC rules: a. b. H-C CH,CH-CH
-
Name the following compounds by the IUPAC system: a. CH3CH=C(CH2CH2CH3)2 b. (CH3)2CHCH"CHCH3 c. g. CH3-C-C-CH-CH, h. k.
-
Name the following compounds and assign oxidation states to the halogens in them: (a) Fe(ClO3)3 (b) HClO2 (c) XeF6 (d) BrF5 (e) XeOF4 (f) HIO3.
-
You are deciding between two mutually exclusive investment opportunities. Both require the same initial investment of $10 million. Investment A will generate $2 million per year (starting at the end...
-
Figure displays the waveforms of four signals s1 (t), s2 (t), s3 (t) and s4 (t). (a) Using the Gram-Schmidt orthogonalization procedure, find an orthonormal basis for this set of signals. (b)...
-
The cross section of a long cylindrical fuel element in a nuclear reactor is shown. Energy generation occurs uniformly in the thorium fuel rod, which is of diameter D = 25 mm and is wrapped in a thin...
-
Water flows through a horizontal 60-mm-diameter galvanized iron pipe at a rate of \(0.02 \mathrm{~m}^{3} / \mathrm{s}\). If the pressure drop is \(135 \mathrm{kPa}\) per \(10 \mathrm{~m}\) of pipe,...
-
Review the opening feature of this chapter dealing with ash&dans and the entrepreneurial owners, Ashley Cook and Danielle Dankner. Required 1. Assume that ash&dans sells a $300 gift certificate to a...
-
Mercier Manufacturing produces a plastic part in three sequential departments: Extruding, Fabricating, and Packaging. Mercier uses the weighted-average process costing method to account for costs of...
-
Name the following compounds.
-
Draw the structure of each of the following molecules. (a) ethyl vinyl ether (b) 2-bromopropanal (c) pentanoic acid (d) 3-fluorophenol
-
A company making tires for bikes is concerned about the exact width of their cyclocross tires. The company has a lower specification limit of 22.8 mm and an upper specification limit of 23.2 mm. The...
-
Explain how you can identify excess motion during an operational audit.
-
Provide three recommendations to minimize the presence of underutilized employees.
-
If inventory is an asset, why is it considered a waste?
-
Describe the impact of defects on organizational success from an internal and external (e.g., competitive) perspective.
-
Which two structures often provide the greatest opportunities for workers to learn from each other thus facilitating the transfer of knowledge laterally in the organization?
-
Julia receives 1,000 shares of Cookery Corporation stock from her grandfather as a wedding present. The shares are selling for $24 per share on the date of the gift. Grandfather paid $8,000 for them...
-
Why is disclosure of depreciation or amortization methods and rates so important?
-
Sunlight incident on a screen containing two long narrow slits 0.20 mm apart casts a pattern on a white sheet of paper 2.0 m beyond. What is the distance separating the violet ( 0 = 400 nm) in the...
-
With regard to Youngs Experiment, derive a general expression for the shift in the vertical position of the mth maximum as a result of placing a thin parallel sheet of glass of index n and thickness...
-
Two narrow slits in a thin metal sheet are 2.70 mm apart centerto-center. When illuminated directly by plane waves (in air) a fringe pattern appears on a screen 4.60 m away. It is found that...
-
How would the answer to part a change if the couple also realize that in 20 years they would need to spend $52,000 on their child's college education?
-
Write Executive Summary'. - Explain what type of stock you selected. - Are they large stocks (large market cap, usually greater than $10 billion)? - Are they growth stocks (high P/E and P/S ratios)...
-
A firm has an initial debt-to-capital ratio of 10% and is planning to get rid of all of its debt. The tax rate is 30% and the firm's initial pre-tax cost of debt is equal to 5%. What is the firm's...

Study smarter with the SolutionInn App


