Set up the equations needed to calculate the pH of a solution that is 0.050 M ammonium
Question:
Set up the equations needed to calculate the pH of a solution that is 0.050 M ammonium ion.
Strategy
The strategy is detailed in the steps listed in the margin.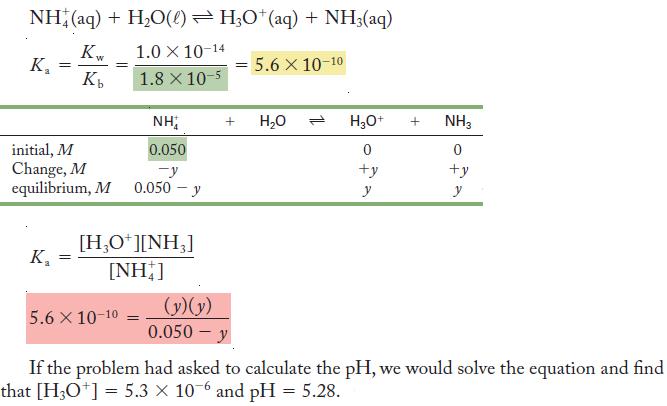
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (2 reviews)
NHaq H2Ol H3O aq NH3aq K Kw Kb 10 x 1014 18 X 105 56 X 10...View the full answer

Answered By

Surojit Das
I have vast knowledge in the field of Mathematics, Business Management and Marketing. Besides, I have been teaching on the topics Management leadership, Business Administration, Human Resource Management, Business Communication, Accounting, Auditing, Organizer Behaviours, Business Writing, Essay Writing, Copy Writing, Blog Writing since 2020. It is my personality to act quickly in any emergency situations when students need my services. I am very professional and serious in every questions students asked me at the time of dealing any projects. I have been serving detailed, quality, properly analysed research paper through the years.
4.80+
91+ Reviews
278+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Calculate the pH of a solution that is 1.00 M HCN and 1.00 M HF. Compare the concentration (in molarity) of the CN2 ion in this solution with that in a 1.00 M HCN solution. Comment on the difference.
-
Calculate the hydronium ion concentration and pH of a solution that is 0.0500 M in HCl (a) Neglecting activities. (b) Using activities.
-
Calculate the pH of a solution that is (a) 0.0100 M in HClO4 and 0.0300 M in monochloroacetic acid. (b) 0.0100 M in HCl and 0.0150 M in H2SO4. (c) 0.0100 M in NaOH and 0.0300 M in Na2S. (d) 0.0100 M...
-
B. Giulia runs at 4.5 m/s (1 m/s is 2.2 mi/h ... just think "about 2 mi/h"). She is running eastward into the sunrise and along a straight-line path. Show a complete sketch from when you start...
-
Is a nucleus that absorbs at 6.50 more shielded or less shielded than a nucleus that absorbs at 3.20 ? Does the nucleus that absorbs at 6.50 require a stronger applied field or a weaker applied...
-
A Chicago-based manufacturer is looking for someone to handle its shipments to the West Coast. In order to evaluate potential transportation providers, the manufacturer has developed the following...
-
Consider the study from Example 3.4. Recall that the clinical trial consists of 450 patients. 150 of the patients have stage I cancer and the rest have stages II-IV cancer. In Computation Lab:...
-
1. In each situation, what are the main financial planning issues that need to be addressed? 2. What additional information would you like to have before recommending actions in each situation? 3....
-
Alexis and Tomas meet with their insurance agent, Pat. After a thorough needs analysis, Pat recommends that Alexis set up a spousal RRSP tot Tornas Pat has them complete a risk tolerance...
-
Calculate the pH of the following solutions. (a) 0.10 M sodium nitrate (b) 0.050 M KF Strategy Look at the ions formed when the salt dissolves. If both are spectator ions, the pH will be set by the...
-
Explain how to calculate the pH of the following solutions: (a) A solution that is 0.25 M KOH and 1.00 M ammonia (b) A solution that is 0.40 M HCl, 0.20 M HBr, 0.10 M HCOOH, and 0.20 M HF (c) A...
-
On March 31, 2018, Southwest Gas leased equipment from a supplier and agreed to pay $200,000 annually for 20 years beginning March 31, 2019. Generally accepted accounting principles require that a...
-
Consideration of Laws and Regulations in an Audit of Financial Statements with examples.
-
Law enforcement needs "probable cause" in order to justify two different acts. What are the two different acts?
-
Discuss the ethical and legal considerations surrounding the use of global variables in software development, including issues related to data privacy, regulatory compliance, and intellectual...
-
Paid fourteen ( 1 4 ) months ' rent on a lease rental contract at $ 3 5 , 0 0 0 . what is the acutal cost per month?
-
why do you believe that the automobile exception exists? Is this a good idea in terms of upholding the spirit of the 4th Amendment?
-
Refer to the Real World Case on Medical IT in the chapter. What do these automated or semi-automated technologies spell for the future of medicine? How much of this discipline can be captured using...
-
A superior criticized a sales manager for selling high-revenue, low-profit items instead of lower-revenue but higher-profit items. The sales manager responded, My income is based on commissions that...
-
The following is the expression for the vector E-field of an electromagnetic wave traveling in a homogeneous dielectric: Here v = 1.80 Ã 10 15 rad/s and k = 1.20 Ã 10 7 rad/m. (a)...
-
A tungsten lightbulb puts out 20 W of radiant energy (most of it IR). Assume it to be a point source and calculate the irradiance 1.00m away.
-
Consider an electromagnetic wave impinging on an electron. It is easy to show kinematically that the average value of the time rate-of-change of the electrons momentum vector p is proportional to the...
-
Melinda brought her Japanese friend, Emiko, home to visit her parents. When Emiko addressed Melinda's mom as "Mrs. Miller," Melinda's mom said, "Oh, please call me Susan." Emiko became very...
-
A customer of your servie experienced a service failure. Your team did an excellent job recovering from the failure, and the customer is very satisfied with the outcome. What does this scenario...
-
Are these goals, strategies or tactics? Use YouTube channel to promote new product offerings, with testimonial support, 2 . Use Instagram posts to create customer engagement & improve brand s...

Study smarter with the SolutionInn App


