The following data were obtained for the decomposition of nitrogen dioxide. 2NO2(g) 2NO(g) + O(g) Time (s)
Question:
The following data were obtained for the decomposition of nitrogen dioxide.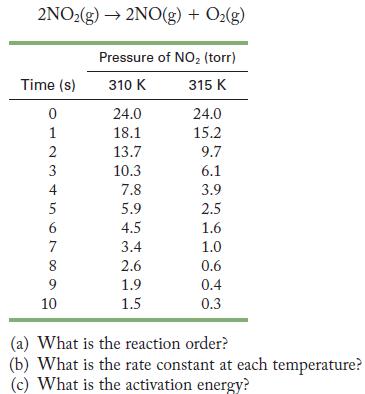
Transcribed Image Text:
2NO2(g) 2NO(g) + O(g) Time (s) 0 1 23456 4 7 8 9 10 Pressure of NO (torr) 310 K 315 K 24.0 18.1 13.7 10.3 7.8 5.9 4.5 3.4 2.6 1.9 1.5 24.0 15.2 9.7 6.1 3.9 2.5 1.6 1.0 0.6 0.4 0.3 (a) What is the reaction order? (b) What is the rate constant at each temperature? (c) What is the activation energy?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
To determine the reaction order rate constant and activation energy we can use the integrated rate l...View the full answer

Answered By

Shameen Tahir
The following are details of my Areas of Effectiveness. The following are details of my Areas of Effectiveness English Language Proficiency, Organization Behavior , consumer Behavior and Marketing, Communication, Applied Statistics, Research Methods , Cognitive & Affective Processes, Cognitive & Affective Processes, Data Analysis in Research, Human Resources Management ,Research Project,
Social Psychology, Personality Psychology, Introduction to Applied Areas of Psychology,
Behavioral Neurosdence , Historical and Contemporary Issues in Psychology, Measurement in Psychology, experimental Psychology,
Business Ethics Business Ethics An introduction to business studies Organization & Management Legal Environment of Business Information Systems in Organizations Operations Management Global Business Policies Industrial Organization Business Strategy Information Management and Technology Company Structure and Organizational Management Accounting & Auditing Financial Accounting Managerial Accounting Accounting for strategy implementation Financial accounting Introduction to bookkeeping and accounting Marketing Marketing Management Professional Development Strategies Business Communications Business planning Commerce & Technology Human resource management General Management Conflict management Leadership Organizational Leadership Supply Chain Management Law Corporate Strategy Creative Writing Analytical Reading & Writing Other Expertise Risk Management Entrepreneurship Management science Organizational behavior Project management Financial Analysis, Research & Companies Valuation And any kind of Excel Queries.
4.70+
16+ Reviews
34+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Choose a logo from a women's professional sports team and a men's professional sports team and complete the following analysis: A1) Paste in the logo for your chosen women's pro sports team and...
-
The following values of the rate constant were obtained for the decomposition of nitrogen dioxide at various temperatures. Plot the logarithm of k versus 1/T and from the graph obtain the energy of...
-
The decomposition of nitrogen dioxide (NO2) into nitrogen monoxide (NO) and oxygen is a second-order reaction. This means that the concentration C of NO2 at time t is given by 1/C = kt + 1/C0, where...
-
The Electronic Industries Association reports that about 50% of U.S. households have a camcorder. For a randomly selected sample of 800 U.S. households, use the normal approximation to the binomial...
-
Find Vx in the circuit infigure. 24 V Vx 4 kn 36 kn 6V(+ +)8 V
-
How do changes in the industry environment affect a businesss risks?
-
Consider the regression model developed for the National Football League data in Problem 3.1. Data From Problem 3.1 Consider the National Football League data in Table B.1. a. Calculate the PRESS...
-
Section 404 of the SarbanesOxley Act requires auditors of a public company to analyze and report on the effectiveness of the clients internal controls over financial reporting. Describe the...
-
What are the key advantages of a deposit account over a money market account? What are the key advantages of a credit line with a bank over commercial paper issuance?
-
The reactant in a first-order reaction decreases in concentration from 0.451 to 0.235 M in 131 seconds. How long does it take to decrease from 0.235 to 0.100 M?
-
Use the following experimental data to determine the rate law and rate constant for formation of phosgene. CO + Cl COC1 Experiment 123 Initial Concentration (M) [CO] 0.053 0.106 0.106 [C] 0.23 0.23...
-
The vapor pressure of water at 40.0C is 7.34 x 10 3 N/m 2 . Using the ideal gas law, calculate the density of water vapor in g/m 3 that creates a partial pressure equal to this vapor pressure. The...
-
As shown in the module resources, the Sarbanes-Oxley Act of 2002 (SOX) has several implications for project management. Describe what you believe is the best use of EVM principle(s) to satisfy SOX...
-
Suppose we wish to set up a production plan for the JC Company for the next six months. We are given the following information: Jan Feb March April May June 22 19 21 21 22 20 2000 1800 1200 1100 1100...
-
Conscious capitalism is reflected in various manners in an organization. How does an organization practice conscious capitalism? How could an organization further its understanding and implementation...
-
An aggregate plan shows inventory at the end of May = 800 units Forecast demand for June = 1200 units Production output expected during June = 1100 units. What is the expected inventory at the end of...
-
1. When brand new technology is introduced in the market, there are no experts. How would you go about trying to decide if technology is a good fit for your business or not? What are concrete steps...
-
You hold a diversified $100,000 portfolio consisting of 20 stocks with $5,000 invested in each. The portfolio's beta is 1.12. You plan to sell a stock with b = 0.90 and use the proceeds to buy a new...
-
Orange juice producers are dismayed and puzzled. An economist told them that the reason the demand for orange juice fell is that a new technology allow tomato producers to pick ripe tomatoes more...
-
Determine the slope and displacement at point B. EI is constant. Using Castiglianos theorem. 400 N 300 N/m 'A 3 m
-
Determine the slope and displacement at point B. EI is constant. Use the method of virtual work. 400 N 300 N/m 'A 3 m
-
Determine the displacement and slope at point C of the cantilever beam. The moment of inertia of each segment is indicated in the figure. Take E = 29(10 3 ). Using Castiglianos theorem. A IBc = 200...
-
When Matthew sends Richard a message, what key should he use to encrypt the message?
-
A perpetuity bond is a coupon bond with infinite maturity. The price-yield relation for the perpetuity bond is P = C/y where P is the price, C the coupon rate and y the yield. Consider two perpetuity...
-
Simplify C 52x-3

Study smarter with the SolutionInn App


