The molecular orbital diagram of NO shown in Figure 10.47 also applies to CO. Draw the complete
Question:
The molecular orbital diagram of NO shown in Figure 10.47 also applies to CO. Draw the complete molecular orbital diagram for CO. What is the C–O bond order?
Figure 10.47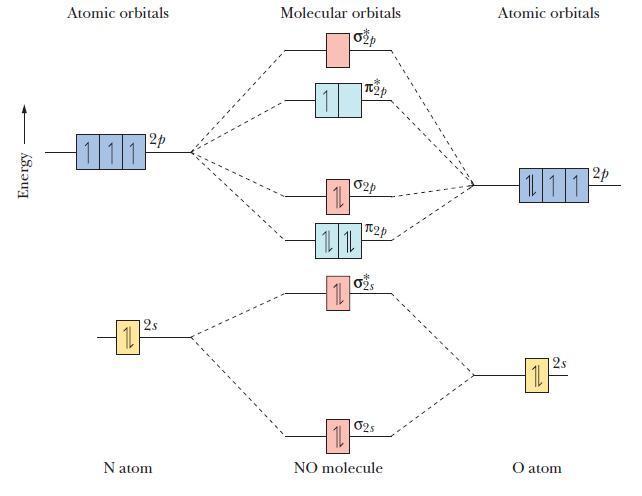
Transcribed Image Text:
Energy Atomic orbitals 12p 111 -10 Natom Molecular orbitals 11 1L 1L 11 12p 02p 12p 02S 025 1L NO molecule Atomic orbitals 111 = 11 2 12p O atom
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
2p 2 s O atom ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
The molecular orbital diagram of NO shown in Figure 10.47 also applies to OF - . Draw the complete molecular orbital diagram for OF - . What is the OF bond order? Figure 10.47 Energy Atomic orbitals...
-
The molecular orbital diagram of NO shown in Figure 10.47 also applies to the following species. Write the molecular orbital electron confi guration of each, indicating the bond order and the number...
-
The nitrosyl ion, NO + , has an interesting chemistry. Assume the molecular orbital diagram shown in Figure 9.16 applies to NO + . (a) Is NO + diamagnetic or paramagnetic? If paramagnetic, how many...
-
Data-2-Go manufactures and sells flash drives. The company produces only when it receives orders and, therefore, has no inventories. The following information is available for the current month:...
-
In the network in figure, the switch opens at t = 0. Use Laplace transforms to find v0(t) for t >0. 4 kn 3 kn 100 F vct): 12 V (+ 2 kn vo(t)
-
The following cumulative frequency polygon shows the selling price ($000) of houses sold in the Billings, Montana, area. a. How many homes were studied? b. What is the class interval? c. One hundred...
-
For the scaling numbers \[\begin{aligned}& \alpha_{1}=\frac{1+\sqrt{3}}{4 \sqrt{2}} \\& \alpha_{2}=\frac{3+\sqrt{3}}{4 \sqrt{2}} \\& \alpha_{3}=\frac{3-\sqrt{3}}{4 \sqrt{2}} \\&...
-
Refer to the preceding facts for Panthers acquisition of Sandin common stock. On January 1, 2012, Panther held merchandise sold to it from Sandin for $12,000. This beginning inventory had an...
-
Explain how the calculations in the the below table are worked out? -the S&P is calculated? -beta -market premium -benchmark -r2 -(1-r2) # Stock Options Weightage Anticaped WT. Avg Rate of Return...
-
Assuming that the molecular orbital diagram shown in Figure 10.40 is correct for heteronuclear diatomic molecules containing elements that are close to each other in the periodic table, write a...
-
Aspartame is a compound that is 200 times sweeter than sugar and is used extensively (under the trade name NutraSweet) in diet soft drinks. The skeleton structure of the atoms in aspartame is H-0. ....
-
The employee benefits manager of a small private university would like to estimate the proportion of full-time employees who prefer adopting the first (i.e., plan A) of three available health care...
-
What role did the general manager(s) of the Golden Apple Restaurant and if any the previous owners Robert and Jasmine Raveendran play in the decline of the current morale and challenges the...
-
Develop an information life cycle for information relating to IT equipment purchased by Match Lighting. Map the use of IT to each event in the life cycle to show how IT can be used to manage the...
-
Choose two businesses within the Healthy Care industry (example Hospital's): 1. Compare and contrast their operations strategy on dimensions such as cost, quality, time, and flexibility. 2. How are...
-
Clayton Christensen, Article: How Will You Measure Your Life? Reflect on his exhortations in the light of your own life. What are your take-aways from this speech as a leader of innovation? The...
-
What are hierarchical plans that address an organization's institutional-level needs and attempt to position it advantageously within its task environment called?
-
You are in charge of a quarry that supplies sand and stone aggregates to your companys construction sites. Empty trucks from construction sites arrive at the quarrys huge piles of sand and stone...
-
Ex. (17): the vector field F = x i-zj + yz k is defined over the volume of the cuboid given by 0x a,0 y b, 0zc, enclosing the surface S. Evaluate the surface integral ff, F. ds?
-
If the load has a weight of 600 lb, determine the maximum normal stress on the cross section of the supporting member at section aa. Also, plot the normal-stress distribution over the cross section....
-
The plate has a thickness of 20 mm and the force P = 3 kN acts along the centerline of this thickness such that d = 150 mm. Plot the distribution of normal stress acting along section aa. 200 mm
-
Determine the maximum distance d to the edge of the plate at which the force P can be applied so that it produces no compressive stresses on the plate at section aa. The plate has a thickness of 20...
-
4 Part A Consider the state of stress on an element at the same point oriented 56.5 counterclockwise with respect to the element shown in (Figure 1). Determine the normal stress on the tilted...
-
e) For a 3-noded triangular element (dimension in mm) in Figure 2d, the displacements (in mm) at the nodes are obtained as [ui vi u2 v2 U3 v3] = [-0.002 0.001 0.00 0.003 0.001 -0.002]. Calculate the...
-
(35 points) The Martian adventures: Mark is running a botanical experiment where he is trying to replicate recent scientific research that tiny electrical 'shocks' given to plants can improve...

Study smarter with the SolutionInn App


