The vapor pressure of water at 25 C is 23.77 torr , increasing to 42.20 torr at
Question:
The vapor pressure of water at 25 °C is 23.77 torr , increasing to 42.20 torr at 35 °C . Calculate the standard free energy and standard enthalpy changes at 25 °C for the vaporization of water.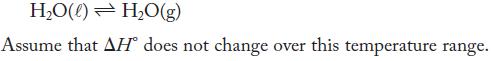
Strategy
First, determine the Gibbs free-energy change of the process; then use the Clausius–Clapeyron equation to determine the enthalpy change.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:





