Use the information in Figure 19.5 to explain why the +3 oxidation state becomes less common for
Question:
Use the information in Figure 19.5 to explain why the +3 oxidation state becomes less common for the elements near the end of the transition series.
Figure 19.5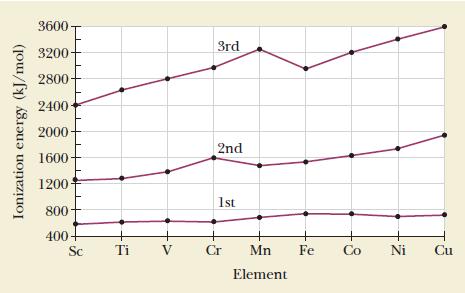
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
The third ioniza...View the full answer

Answered By

Asim farooq
I have done MS finance and expertise in the field of Accounting, finance, cost accounting, security analysis and portfolio management and management, MS office is at my fingertips, I want my client to take advantage of my practical knowledge. I have been mentoring my client on a freelancer website from last two years, Currently I am working in Telecom company as a financial analyst and before that working as an accountant with Pepsi for one year. I also join a nonprofit organization as a finance assistant to my job duties are making payment to client after tax calculation, I have started my professional career from teaching I was teaching to a master's level student for two years in the evening.
My Expert Service
Financial accounting, Financial management, Cost accounting, Human resource management, Business communication and report writing. Financial accounting : • Journal entries • Financial statements including balance sheet, Profit & Loss account, Cash flow statement • Adjustment entries • Ratio analysis • Accounting concepts • Single entry accounting • Double entry accounting • Bills of exchange • Bank reconciliation statements Cost accounting : • Budgeting • Job order costing • Process costing • Cost of goods sold Financial management : • Capital budgeting • Net Present Value (NPV) • Internal Rate of Return (IRR) • Payback period • Discounted cash flows • Financial analysis • Capital assets pricing model • Simple interest, Compound interest & annuities
4.40+
65+ Reviews
86+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
Use the information in Figure 32.1A and 32.1C to answer the following questions: a. What is the six-month forward rate for the Japanese yen, in yen per Canadian dollar? Is the yen selling at a...
-
A 110 g hockey puck sent sliding over ice is stopped in 15 m by the frictional force on it from the ice. (a) If its initial speed is 6.0m/s, what is the magnitude of the frictional force? (b) What is...
-
Write down logical representations for the following sentences, suitable for use with Generalized Modus Ponens: a. Horses, cows and pigs arc mammals. b. An offspring of a horse is a horse. c....
-
The rectangular platform is hinged at A and B and is supported by a cable that passes over a frictionless hook at E. Knowing that the tension in the cable is 1349 N, determine the moment about each...
-
The plate shown in Fig. P5.56 is \(0.5 \mathrm{~m}\) wide perpendicular to the paper. Calculate the velocity of the water jet required to hold the plate upright. Figure P5.56 d = 10.0 cm 20C water...
-
Ludlow Plastics manufactures custom park furniture and signage from recycled plastics (primarily shredded milk jugs.) Many of the companys customers are municipalities that are required by law to...
-
Bond X is a premium $1000 par value bond making annual payments. The bond has a coupon rate of 9%, a YTM of 7%, and has 13 years to maturity. Bond Y is a discount $1000 par value bond making annual...
-
Name and sketch the four most important shapes of transition-metal complexes.
-
Why do the atomic radii of the transition elements within a period decrease more rapidly from Group 3B through 6B than through the rest of the transition elements in that period?
-
A sample of difference scores from a repeatedmeasures experiment has a mean of MD = 4 with a standard deviation of = 6. a. If n = 4, is this sample sufficient to reject the null hypothesis using a...
-
In 1911 the Supreme Court decided to ________. a) allow the trusts to keep functioning as they had in the past b) break up the trusts c) let the trusts off with small fi nes d) put the leaders of the...
-
The monopolistic competitor _________ produces at the minimum point of his or her ATC curve.
-
Joe Spivey is president of Advantage Research, Inc. The firm specializes in customized research for clients in a variety of industries via computer-assisted mall interviews in five of the largest...
-
Using Gauss elimination and back substitution, solve 8 2 1 4 6 2 3 4 14 X X 3 4 2
-
In 1911 the Supreme Court broke up the______________- and the _______________.
-
In general, a taxpayer can exclude up to $250,000 of gain on the sale of a principal residence. However, this exclusion is only available every two years. Explain the circumstances under which the...
-
The cash records of Holly Company show the following four situations. 1. The June 30 bank reconciliation indicated that deposits in transit total $720. During July, the general ledger account Cash...
-
In sand derived from the attack of waves on granite, what mineral would you expect to be most abundant?
-
Distinguish between an alluvial fan and a moraine.
-
What is the immediate destination of most of the water that falls as rain on land?
-
1. Consider a market in which the Capital Asset Pricing Model (CAPM) holds. There are two assets with the following attributes.: Rate of return (p.a.) State Probability Asset 1. Asset 2 1 0.2 5% 11%...
-
How can you balance innovation and stability in healthcare management?
-
In what way(s) do you feel your body language was interpreted by the audience?

Study smarter with the SolutionInn App


