Use the solubility rules in Table 4.1 to predict whether BaSO 4 is soluble or insoluble in
Question:
Use the solubility rules in Table 4.1 to predict whether BaSO4 is soluble or insoluble in water.
Table 4.1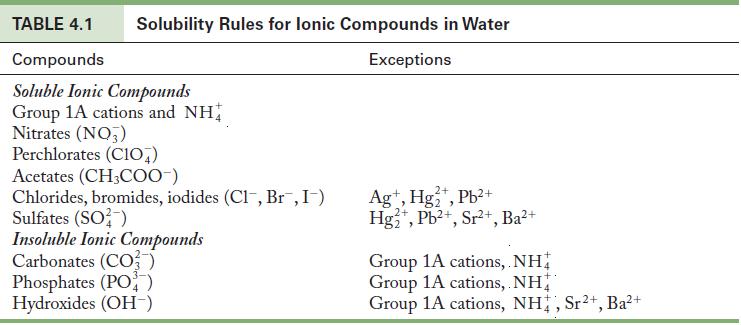
Transcribed Image Text:
TABLE 4.1 Compounds Soluble Ionic Compounds Group 1A cations and NH Nitrates (NO3) Solubility Rules for lonic Compounds in Water Exceptions Perchlorates (C107) Acetates (CH3COO-) Chlorides, bromides, iodides (Cl, Br¯, I¯) Sulfates (SO²) Insoluble Ionic Compounds Carbonates (CO3) Phosphates (PO4) Hydroxides (OH-) 2+ Ag+, Hg₂+, Pb²+ Hg2+, Pb²+, Sr²+, Ba²+ Group 1A cations, NH Group 1A cations, NH Group 1A cations, NH, Sr²+, Ba²+
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
BaS...View the full answer

Answered By

Jonas Araujo
I have recently received the degree of PhD. In Physics by the Universidade Federal do Maranhão after spending a term in Durham University, as I have been awarded a scholarship from a Brazilian mobility program. During my PhD. I have performed research mainly in Theoretical Physics and published works in distinguished Journals (check my ORCID: https://orcid.org/0000-0002-4324-1184).
During my BSc. I have been awarded a scholarship to study for a year in the University of Evansville, where I have worked in detection-analysis of photon correlations in the the Photonics Laboratory. There I was a tutor in Electromagnetism, Classical Mechanics and Calculus for most of that year (2012).
I am very dedicated, honest and a fast learner, but most of all, I value a job well done.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Using the solubility rules in Table 4.1 as a guide, predict whether an insoluble product forms when each of the following pairs of solutions is mixed. Write the balanced chemical equation if a...
-
Are the compounds listed below soluble or insoluble in water? (a) Ba(NO 3 ) 2 (b) PbSO 4 (c) LiOH (d) AgCl Strategy Use the solubility rules in Table 4.1 to predict whether each compound is soluble...
-
Part 1 a. Ammonia, NH 3 , is a weak electrolyte. It forms ions in solution by reacting with water molecules to form the ammonium ion and hydroxide ion. Write the balanced chemical reaction for this...
-
On the accompanying graph, draw the consumption function C = $200 + 0.75YD. (a) At what level of income do households begin to save? Designate that point on the graph with the letter A. (b) By how...
-
A small spherical satellite is in circular orbit round the Sun. The Sun subtends at the satellite a solid angle of 7 105 steradians, and the temperature of the satellite is uniform. Assuming that...
-
Let G be a nonempty finite set with an associative binary operation such that for all a, b, c G ab= ac b = c and ba = ca b = c. Then G is a group. Show that this conclusion may be false if G is...
-
A negatively charged particle located at the origin of an \(x y z\) coordinate system is spinning clockwise about the \(x\) axis (that is, clockwise when you look at the particle while standing...
-
Three different plans for financing a $5,000,000 corporation are under consideration by its organizers. Under each of the following plans, the securities will be issued at their par or face amount,...
-
1. What is a data structure? 2. Why do we need data structures? 3. List some common data structures. 4. How data structures are classified? 5. Differentiate linear and non-linear data structure.
-
BUSINESS CASE (100 points) Julia has recently opened a dry fruits wholesale company dedicated to the sale of peanuts, almonds and pistachios. The company's name is "The Nuthouse". THE NUTHOUSE Since...
-
Th e reaction of acetylene, C 2 H 2 , with oxygen, O 2 , yields carbon dioxide, CO 2 , and water. Write the balanced chemical equation for this reaction. As shown, this reaction produces a very hot...
-
A solution is formed by dissolving 3 g sugar in 100 mL water. Identify the solvent and the solute.
-
Youve observed the following returns on Yamauchi Corporations stock over the past five years: -10 percent, 24 percent, 21 percent, 11 percent, and 8 percent. a. What was the arithmetic average return...
-
An individual has the genotype Aa Bb Cc and makes an abnormal gamete with the genotype AaBc. Does this gamete violate the law of independent assortment or the law of segregation (or both)? Explain...
-
What is cDNA? How does cDNA differ from genomic DNA in eukaryotes?
-
Would epigenetic changes that promote cancer be passed to offspring?
-
With regard to genotypes, what is a true-breeding organism?
-
When a chi square test is applied to solve a linkage problem, explain why an independent assortment hypothesis is proposed.
-
Job Inc. owns 20,000 shares of No job Company which it bought in 2005. No job's total stockholders' equity is as follows: Common stock , $20 par ............$500,000 Additional paid in capital...
-
Troy is a qualified radiologist who operates a successful radiology practice from purpose- built rooms attached to his house. Troy works in the practice three days a week, and the other two days he...
-
Lindane (hexachlorocyclohexane) is an agricultural insecticide that can also be used in the treatment of head lice. Draw both chair conformations of lindane. CI CI 'CI CI I Lindane
-
Draw the lowest energy conformation for each of the following compounds: (a) (b) (c) (d) (e) (f) .CI 'CI
-
You drew the two chair conformations of lindane. Carefully inspect them, and predict the difference in energy between them, if any.
-
4-34. A crate of weight W = 100 lb sits on a ramp oriented at 27 degrees relative to ground, as shown in Fig. P4.34. The free-body diagram showing the external forces is also shown in Fig. P4.34. a....
-
Assume that the following quote for the Walt Disney Company, a NYSE stock, appeared on some particular date of 2019 on Yahoo! Finance (https://finance.yahoo.com/quote/DIS?p=DIS&.tsrc-fin-srch): The...
-
Without a microphone, a certain singer in the concert hall produces 67 dB. With a microphone, this increases to 83.5 dB. a) The sound intensity with the microphone is how many times the previous...

Study smarter with the SolutionInn App


