What is the formula of the molecule pictured below? Carbon Hydrogen
Question:
What is the formula of the molecule pictured below?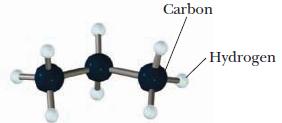
Transcribed Image Text:
Carbon Hydrogen
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
C...View the full answer

Answered By

Raunak Agarwal
Teaching is my hobby and now my profession. I teach students of CA and CFA(USA) in batches of 100 students and have a 5 year experience.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
The molecule pictured below is epinephrine, a compound used as a bronchodilator and antiglaucoma agent. (a) Give a value for each of the indicated bond angles. (b) What are the most polar bonds in...
-
The unusual molecule [2.2.2] propel-lane is pictured in the margin. On the basis of the given structural parameters, what hybridization scheme best describes the carbons marked by asterisks? (Make a...
-
Catalytic cracking is an industrial process used to convert high-molecular-mass hydrocarbons to low-molecularmass hydrocarbons. A petroleum company has a huge supply of heating oil stored as...
-
What are the "two main lines" of distortion of the view of Marx and what does Lenin think the proper view?
-
(a) What is a statement of cash flows? (b) John Norris maintains that the statement of cash flows is an optional financial statement. Do you agree? Explain.
-
Use polar coordinates to describe the region shown. -4 -2 2 -2 y 2 4 X
-
This problem extends the Summary Problem 1 to a second department. During May, Florida Tile Industries reports the following in its Tile-Finishing Department: Requirements: 1. Complete the five-step...
-
Two items are omitted from each of the following summaries of balance sheet and income statement data for two proprietorships for the year 2012, Gavin??s Goods and Floyd...
-
The price of a car you want is $39,000 today. Its price is expected to increase by $1000 each year. You now have $23,500 in an investment account, which is earning 11% per year. How many years will...
-
Identify an element that fits the following criteria: (a) What element in the fourth period is an alkaline earth metal? (b) What element in the second period is a halogen? Strategy Use the periodic...
-
How does the nuclear model of the atom explain the results of the Rutherford experiment?
-
The Rio Olympics 2016 once again proved that major sporting events would struggle to attract the necessary finance and backing if it were not for the brands that sponsor them. Brand owners know that...
-
You currently hold a portfolio of three stocks, Delta, Gamma, and Omega. Delta has a volatility of 44%, Gamma has a volatility of 47%, and Omega has a volatility of 48%. Suppose you invest 30% of...
-
Consider an equally weighted portfolio of stocks in which each stock has a volatility of 40%, and the correlation between each pair of stocks is 27%. a. What is the volatility of the portfolio as the...
-
XL Sports is expected to generate free cash flows of $11.2 million per year. XL has permanent debt of $35 million, a tax rate of 37%, and an unlevered cost of capital of 10.4%. a. What is the value...
-
What are the pluses and minuses of corporate location subsidies? Why do politicians like them so much? Would you be surprised to know that many of the 238 cities bidding for Amazons HQ2 offered much...
-
Does traditional one-person-one-vote (1p1v) majority voting allow voters to directly express differences in strengths of preference? Does quadratic voting do any better? Discuss the differences and...
-
What is usually the biggest expense item for a bank?
-
Solve the relation Exz:Solve therelation ne %3D
-
Predict the products for each of the following Diels-Alder reactions: (a) (b) (c) (d) (e) (f) . C CN
-
Predict the products for each of the following Diels-Alder reactions: (a) (b) (c) (d) (e) (f) HOOC, . COOH
-
Identify the reagents you would use to prepare each of the following compounds via a Diels-Alder reaction: (a) (b) (c) (d) (e) (f) (g) (h) COOH COOH
-
Question 20 (1 point) Common types of taxes one pays in Canada are: Question 20 options: GST, HST & PST Property Tax Income Taxes All of the above
-
you retire with a large amount of savings in aretirement account. You decide to withdraw 3.5% to 4% of your savings the first year and increase the rate by inflation each year. What is the basis of...
-
test 1 for financial analysis and budgeting 1013 lambton college fpwt

Study smarter with the SolutionInn App


