When methyl bromide reacts with hydroxide ion, methyl alcohol and bromide ion form. CH3Br + OH CH3OH
Question:
When methyl bromide reacts with hydroxide ion, methyl alcohol and bromide ion form.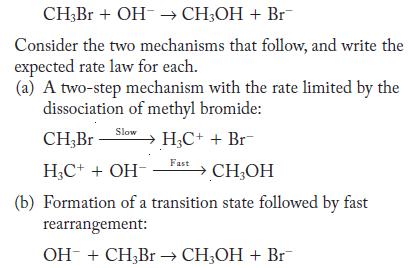
Transcribed Image Text:
CH3Br + OH CH3OH + Br Consider the two mechanisms that follow, and write the expected rate law for each. (a) A two-step mechanism with the rate limited by the dissociation of methyl bromide: Slow CH,Br HC++ Br HC+ + OH- (b) Formation of a transition state followed by fast rearrangement: OH + CH3Br CH3OH + Br Fast CHOH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)
You are correctThe reaction between methyl bromide CHBr and hydroxide ion OH is a classic example of a nucleophilic substitution reactionwhere the hyd...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
When methyl bromide reacts with hydroxide ion in solution, methyl alcohol and bromide ion form. Determine the rate law and evaluate the rate constant from the experimental data. Strategy An...
-
When magnesium metal is burned in air (Figure 3.6), two products are produced. One is magnesium oxide, MgO. The other is the product of the reaction of Mg with molecular nitrogen, magnesium nitride....
-
The reaction between ethyl bromide (C2H5Br) and hydroxide ion in ethyl alcohol at 330 K, C2H5Br(alc) + OH (alc) -- C2H5OH(I) + Br(aIc), is first order each in ethyl bromide and hydroxide ion. When...
-
Explain why entrepreneurial firms are often in a strong position to use combination strategies.
-
Find V1 in the network infigure. Vx + ww 10 kn 5 kn V1 +)25 V +1
-
Conduct additional research and provide evidence in support of or refuting the following statement: All forms of gene therapy used today result in heritable changes.
-
In Problem 3.5 a regression model was developed for the gasoline mileage data using the regressor engine displacement \(x_{1}\) and number of carburetor barrels \(x_{6}\). Calculate the PRESS...
-
High Range produces fleece jackets. The company uses JIT costing for its JIT production system. High Range has two inventory accounts: Raw and In-Process Inventory and Finished Goods Inventory. On...
-
Suppose that zero interest rates with continuous compounding are as follows: Matury (in years) Rate (% per annum) Maturity Rate % 1 3..5 2 4.0 3 4.2 4 ...
-
Ethyl chloride decomposes to form ethylene and hydrogen chloride at 437 K. The reaction takes place in a 4.0-L container and is monitored by measuring the time needed for the hydrogen chloride to...
-
Two reactions have activation energies of 45 and 40 kJ/mol, respectively. Which reaction shows the greater increase in rate with an increase in temperature?
-
Give an example of a contingency table for which the chi-squared test of independence should not be used.
-
Better Mousetraps has developed a new trap. It can go into production for an initial investment in equipment of $5.4 million. The equipment will be depreciated straight-line over 6 years, but, in...
-
How can practitioners ensure that the data they collect is accurate and representative of the organization? What are the challenges that practitioners face when collecting data and how can they...
-
The Toyota production system advocates holding some WIP between consecutive stations in the production process (this is implemented through the Kanban system). Explain the purpose of this inventory...
-
questions. 1. find out the meaning of the Foreign Corrupt Practices Act (FCPA) Why is it illegal and unethical to bribe a foreign official? 2. Search online for "Corruption perception index...
-
Inventory management and aggregate planning serve an obvious need in production companies, but these concepts are also highly valuable for service businesses. Identify an existing service based...
-
Grocer Services Corporation (a calendar year taxpayer), a wholesale distributor of food, made the following donations to qualified charitable organizations during the year: Adjusted Basis Fair Market...
-
Maria Castigliani is head of the purchasing department of Ambrosiana Merceti, a medium-sized construction company. One morning she walked into the office and said, The main problem in this office is...
-
The L-shaped frame is made from two segments, each of length Land flexural stiffness EI. If it is subjected to the uniform distributed load, determine the vertical displacement of point B. Using...
-
The L-shaped frame is made from two segments, each of length Land flexural stiffness EI. If it is subjected to the uniform distributed load, determine the horizontal displacement of the end C. Use...
-
Determine the vertical deflection at C. The cross-sectional area and moment of inertia of each segment is shown in the figure. Take E = 200 GPa. Assume A is a fixed support. Including the effect of...
-
Divide 8(cos0.8 + i sin0.8) by 4(cos0.2 + i sin0.2).
-
Ursula is seeking to expand the reach and scalability of her organization's website. She would like to position copies of her data around the world in locations close to website visitors to reduce...
-
How has the Coronavirus affected education Worldwide? Explain

Study smarter with the SolutionInn App


