Write a balanced equation for each of these combustion reactions. (a) C4H10(g) + O(g) (b) C6H12O6(s)
Question:
Write a balanced equation for each of these combustion reactions.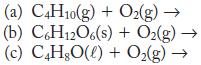
Transcribed Image Text:
(a) C4H10(g) + O₂(g) → (b) C6H12O6(s) + O₂(g) → (c) C4H8O(l) + O₂(g) →>>
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
balanced chemical equations for each of these combustion reactions a C...View the full answer

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Write a balanced equation for each reaction. (a) (b) (c) (d) H SO, heat CH3 CH2CH-CH NaOC(CH3 3 Br Br Nal CHCH CH-CH acetone NaOH, heat CH3 CH CCH3 Br
-
Write a balanced equation for the combustion of benzoic acid, C6H5CO2H, to give CO2 and H2O. How many milligrams of CO2 and of H2O will be produced by the combustion of 4.635 mg of benzoic acid?
-
Write a balanced equation for each of the following reactions or reaction sequences. (a) The reaction catalyzed by PFK-2 (b) The conversion of 2 moles of oxaloacetate to glucose (c) The conversion of...
-
An interest payment of $650 in a 20 percent tax bracket would result in a tax savings of _____.
-
A transmission line is terminated by a load with admittance YL = (0.6 4 + j0.8)/Zo. Find the normalized input impedance at /6 from the load.
-
Rectangle A has a height of (x + 1) meters and an area of 4 m. Rectangle B has a height of (2x 1) meters and an area of 6 m. The sum of the widths of the two rectangles is less than 8 meters. Find...
-
A business firm is thinking of buying a capital good, which will earn \($2,000\) a year for the next four years and cost \($7,000.\) The interest rate is 8 percent. Should the firm buy the capital...
-
Thomas Rinks and Joseph Shields developed Psycho Chihuahua, a caricature of a Chihuahua dog with a do-not-back-down attitude. They promoted and marketed the character through their company, Wrench,...
-
11 12 13 14 Height of a plant is regulated by: a) DNA which is directly influenced by growth hormone. b) Genes which regulate the proteins directly. Growth hormones under the influence of the enzymes...
-
Paul and his wife Gail jointly own (50% each) a piece of vacant land north of Toronto. This land was purchased on January 1, 2017 at a cost of $500,000. On the purchase they paid $25,000 in legal...
-
Write a balanced equation for the combustion (in excess oxygen) of each of the following compounds. (a) C 6 H 12 (b) C 4 H 8 (c) C 2 H 4 O (d) C 4 H 6 O 2
-
Write a balanced equation for the reaction of (a) Mg(OH) 2 and HF. (b) Sodium hydroxide and HCl. (c) H 2 SO 4 and strontium hydroxide.
-
A hypothetical AB alloy of composition 55 wt% B45 wt% A at some temperature is found to consist of mass fractions of 0.5 for both and phases. If the composition of the phase is 90 wt% B10 wt% A,...
-
Compute generalized least squares estimates of the linear probability model.
-
Explain the nature of regressions that involve lagged variables and the number of observations that are available.
-
Compute heteroskedasticity-consistent standard errors for least squares.
-
Test for heteroskedasticity using a Goldfeld-Quandt test applied to (a) two subsamples with potentially different variances and (b) a model where the variance is hypothesized to depend on an...
-
Use a Lagrange multiplier test for serially correlated errors.
-
a. Assume the interest rate in the market (yield to maturity) goes down to 8 percent for the 10 percent bonds. Using column 2, indicate what the bond price will be with a 5-year, a 15-year, and a...
-
Which of the following statements is false? a. Capital leases are not commonly reported in a Capital Projects Fund. b. A governmental entity may report a Capital Project Fund in one year but not the...
-
Calculate O2 mixture (298 15 K, 1 bar) , for oxygen in air, assuming that the mole fraction of O 2 in air is 0.210. Use the conventional molar Gibbs energy defined in Section 6.17.
-
Predict the product of the following reaction. Br NaOCH3, heat -NO2
-
What is the relationship between the K P for the two reactions 3/2H 2 (g) + 1/2N 2 (g) NH 3 (g) and 3H 2 (g) + N 2 (g) 2NH 3 (g)?
-
(a) The energy in an infinite string with no external forces is given by E(1)=(Sw + Tw) dx. Show 0 and hence E(t) = E(0). You may assume that lim, >100 x = 0. (b) If show E(t)=T. y(x, 0) = f(x) = {o...
-
Letter Assignment For this assignment, I would like you to pretend that you are the coordinator for an upcoming trade show that will be held at the Columbus, OH Convention Center. You have received a...
-
How do leaders navigate the complexities of team dynamics, including conflicts, power struggles, and role ambiguity, while promoting constructive dialogue, conflict resolution, and mutual...

Study smarter with the SolutionInn App


