Write all important resonance structures for each of the following species, and use the VSEPR model to
Question:
Write all important resonance structures for each of the following species, and use the VSEPR model to predict the bond angles around each central atom. Also indicate the hybrid orbitals on each central atom and whether the molecule is polar or nonpolar. Does each resonance structure use the same hybrid orbitals?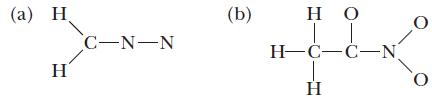
Transcribed Image Text:
(a) C-N-N (b) IT HC-C-N
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
Lets analyze the species given in the image step by step starting with compound a then moving on to compound b Compound a H2CNN 1 Resonance Structures For compound a we are looking at a diazomethane m...View the full answer

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Write all important resonance structures for each of the following species, and use the VSEPR model to determine the bond angles around each central atom. Also indicate the hybrid orbitals on each...
-
Write one important resonance structure for each of the following species, and use the VSEPR model to predict the bond angles around each central atom. Also indicate the hybrid orbitals on each...
-
Write the important resonance structures for each of the following: (a) (b) (c) (d) (e) (f) (g) (h) (i) (j) CH2 CH-Br NO2
-
Arginine, the most basic of the 20 common amino acids, contains a guanidino functional group in its side chain. Explain, using resonance structures to show how the protonated guanidino group...
-
Net Fixed Assets and Depreciation On the balance sheet, the net fixed assets (NFA) account is equal to the gross fixed assets (FA) account (which records the acquisition cost of fixed assets) minus...
-
American Sports, an outdoor sports retailer, is planning to add a Web site for online sales. The estimated costs of two alternative approaches are as follows: At the expected level of orders, which...
-
Differentiate technology from methodology and from method. Can you come up with an example that differentiates these concepts in a specific context, perhaps software development?
-
Multiple Choice Questions The auditor would send a bank confirmation to all banks with which the client had business during the year, because a. The confirmation seeks information on indebtedness...
-
Discuss three social and legal concerns for the internet and e-business. Be specific and provide examples
-
Ionization energies can be determined for molecules and atoms. Draw the molecular orbital diagrams for NO and CO, and predict which compound has the lower ionization energy.
-
More than 5 billion pounds of ethylene oxide, C 2 H 4 O, is produced annually. Ethylene oxide is used in the production of ethylene glycol, HOCH 2 CH 2 OH, the main component of antifreeze, and...
-
End A of rod AB moves to the right with a constant velocity of 6 ft/s. For the position shown, determine (a) the angular acceleration of rod AB, (b) the acceleration of the midpoint G of rod AB. 4 ft...
-
The Line class (2.5 pts) The Line class represents a 2D line that stores two Point2D objects and provides the distance between the two points and the slope of the line using the property methods...
-
Given the Flight Centre monthly share prices from Jan 2020 - Dec 2020, calculate the monthly returns, and the standard deviation. (3.5 marks) [Ensure to show your workings. Percentage results must be...
-
Develop a program to compute N! using unsigned integer type of array to store; for a N from 1 to 99,999,999. The result can be a very long number. For example, the factorial of 50 is:...
-
Objective: This assignment aims to deepen students' understanding of application security by analyzing real-world case studies. Students will explore how vulnerabilities were exploited, the impacts,...
-
Tatiana explains to her grandmother (Mamie) how, now with the Internet, things have changed. Complete their conversation with the correct form of the verbs in parentheses. Tatiana: Now, we no longer...
-
The Ajax International Company operates four factories that ship products to five warehouses. The shipping costs requirements and capacities are shown in Figure. Use the transportation method to find...
-
Using the information presented in Problem 13.4B, prepare a partial statement of cash flows for the current year, showing the computation of net cash flows from operating activities using the...
-
If the allowable bending stress is Ï allow = 6 MPa, determine the minimum dimension d of the beams crosssectional area to the nearest mm. 125 mm 25 mm / 25 mm 75 mm 12 kN 8 kN/m 75 mm B- 2 m- 4m
-
The beam has a rectangular cross section as shown. Determine the largest intensity w of the uniform distributed load so that the bending stress in the beam does not exceed Ï max = 10 MPa. 50 mm...
-
The beam has the rectangular cross section shown. If w = 1 kN/m, determine the maximum bending stress in the beam. Sketch the stress distribution acting over the cross section. 50 mm I T150 mm -2 2...
-
Pro Forma Income Statement Sales CMR Company without Project Potential Project $ 1,000,000 $ 70% Company with Project 200,000 $ 60% 1,200,000 Variable Expenses Contribution Margin Fixed Expenses $...
-
Complete the definitions of the quantifiers below. (2) has been filled in as an example. (2) [More-than-six x: F(x)] G(x) is true iff |FG|> 6 (As in More than six seals barked. In each of the example...
-
Explain the importance and value of HR services and strategies within an organisation

Study smarter with the SolutionInn App


