Write balanced equations for the following reactions. (a) C5H12 + O CO + HO (b) NH3
Question:
Write balanced equations for the following reactions.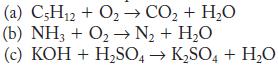
Transcribed Image Text:
(a) C5H12 + O₂ →→ CO₂ + H₂O (b) NH3 + O₂ → N₂ + H₂O (c) KOH + H₂SO4 → K₂SO4 + H₂O
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
a C 5 H 12 8O 2 5C...View the full answer

Answered By

HARSH RANJAN
Taken classes at college to graduates, Also worked as an expert to a freelancer online question-solving portal for more than 8 months with an average rating greater than 4.2 out of 5.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Write balanced nuclear equations for the following reactions and identify X:
-
Write balanced nuclear equations for the following reactions and identify X: 80 34
-
Write balanced equations for each of the following reactions (some of these are analogous to reactions shown in the chapter). (a) Aluminum metal reacts with acids to form hydrogen gas. (b) Steam...
-
1. Is religious training mandatory at all of the schools in question? 2. Does the Court see inevitable church-state entanglements if the Board were allowed to exercise jurisdiction over teachers in...
-
Refer to the lossless transmission line shown in Figure.(a) Find T and s.(b) Determine Zin at the generator. ....... 120 2 Z, = 50 2 Vg
-
Find f 1 (x), stating its domain. f(x) = 1 + 2x/x 1, x R, x 1.
-
In 2020, the Bureau of Labor Statistics posted the following statement to its website: Due to the unusual circumstances related to the pandemic . . . interviewers were given additional guidance...
-
Enviro-Tech has only two retail and two wholesale customers. Information relating to each customer for 2013 follows (in thousands): Enviro-Techs annual distribution-channel costs are $ 33 million for...
-
26 The number of protons, neutrons, electrons in some particles are shown in the table below Particle Protons Neutrons electrons P 1 1 2 Q 2 2 2 R 3 4 2 T 4 5 4 Which one of the following particles...
-
On January 1, 2010, Porter Company purchased an 80% interest in the capital stock of Salem Company for $850,000. At that time, Salem Company had capital stock of $550,000 an retained earnings of...
-
What is the percent yield if 2.4 g NH 3 is obtained from the reaction of 0.64 g H 2 with excess N 2 ?
-
A mixture of sulfur dioxide and oxygen gas reacts as shown below. (a) Write the balanced equation (remember to express the coefficients as the lowest set of whole numbers). (b) Name the product....
-
What are financial markets? What function do they perform? How would an economy be worse off without them?
-
Rank these atoms by the number of electrons they tend to lose, from fewest to most: (a) sodium, Na; (b) magnesium, Mg; (c) aluminum, Al.
-
Why does it make sense to think that the alcohol is made of very tiny particles (molecules) rather than being an infinitely continuous material?
-
Generally, what types of changes in desired saving and desired investment lead to large current account deficits in a small open economy? What factors lead to these changes in desired saving and...
-
Why does it take so much more energy to boil 10 grams of liquid water than to melt 10 grams of ice?
-
Rank these noble-gas atoms by their number of filled shells, from least to most: (a) argon, (b) radon, (c) helium, (d) neon.
-
El Capitan Foods has a capital structure of 40% debt and 60% equity, its tax rate is 35%, and its beta (leveraged) is 1.25. Based on the Hamada equation, what would the firm's beta be if it used no...
-
Consider the advantages and disadvantages of extending property rights so that everyone would have the right to prevent people imposing any costs on them whatsoever (or charging them to do so).
-
Under what conditions is the distribution of products in an ideal gas reactions system at equilibrium unaffected by an increase in the pressure?
-
Identify which of the following compounds is most activated toward electrophilic aromatic substitution. Which compound is least activated? Br NO2 NO2 OMe .
-
Predict the product(s) obtained when each of the following compounds is treated with a mixture of nitric acid and sulfuric acid: (a) (b) (c) (d) (e) Br
-
Owners of a car rental company have determined that if they charge customers p dollars per day to rent a car, where 50 p 200, the number of cars n they rent per day can be modeled by the linear...
-
A 24.8-mW laser beam of diameter 1.86 mm is reflected at normal incidence by a perfectly reflecting mirror. Calculate the radiation pressure on the mirror. N/m
-
a) Suppose that today you take a short position on 10 futures contracts to sell product A in 6 months from now. Each contract is for delivery of 250 pounds of the underlying asset. The futures price...

Study smarter with the SolutionInn App


