Write the chemical equation and use the data in Tables 15.6 and 15.8 to calculate the acid
Question:
Write the chemical equation and use the data in Tables 15.6 and 15.8 to calculate the acid ionization constant for the following ions.
(a) Pyridinium ion
(b) Hydrazinium ion
Table 15.6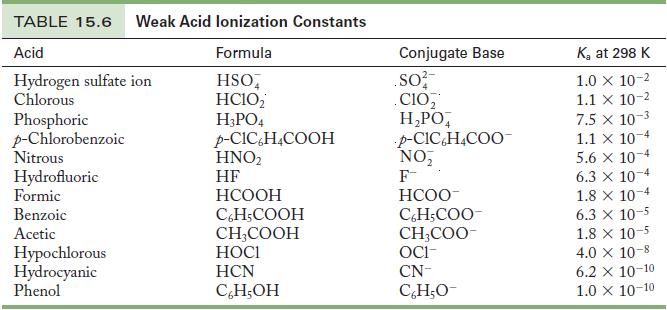
Table 15.8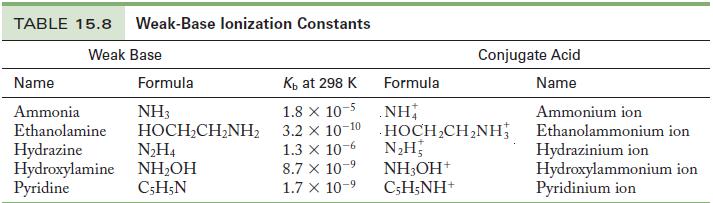
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
Answer Acid Ionization Constants of Pyridinium and Hydrazinium Ions a Pyridinium Ion C5H5NH Pyridini...View the full answer

Answered By

Ali Khawaja
my expertise are as follows: financial accounting : - journal entries - financial statements including balance sheet, profit & loss account, cash flow statement & statement of changes in equity -consolidated statement of financial position. -ratio analysis -depreciation methods -accounting concepts -understanding and application of all international financial reporting standards (ifrs) -international accounting standards (ias) -etc business analysis : -business strategy -strategic choices -business processes -e-business -e-marketing -project management -finance -hrm financial management : -project appraisal -capital budgeting -net present value (npv) -internal rate of return (irr) -net present value(npv) -payback period -strategic position -strategic choices -information technology -project management -finance -human resource management auditing: -internal audit -external audit -substantive procedures -analytic procedures -designing and assessment of internal controls -developing the flow charts & data flow diagrams -audit reports -engagement letter -materiality economics: -micro -macro -game theory -econometric -mathematical application in economics -empirical macroeconomics -international trade -international political economy -monetary theory and policy -public economics ,business law, and all regarding commerce
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Is the criterion 6 3CO 2 ) 2 (aq) is found to be 9.11. However, the contribution to the pH from the autoprotolysis of water was ignored. Repeat the calculation of the pH of this solution, taking into...
-
Calcium acetate, Ca(CH 3 CO 2 ) 2 (aq), is used to treat patients with a kidney disease that results in high levels of phosphate ions in the blood. The calcium binds to the phosphates so that they...
-
Although there are extensive tables available for the pK a of weak acids, you might be dealing with an unknown acid or a known acid at an unlisted temperature. You could then use a procedure like...
-
A. What is Alpha One's NOPAT? Why does NOPAT differ from the earnings after taxes? B. Estimate the effective before-tax cost of the long-term debt. c. Estimate the effective after-tax cost of the...
-
Hot exhaust gases are used in a shell-and-tube exchanger to heat 2.5 kg/s of water from 35 to 85C. The gases assumed to have the properties of air, enter at 200C and leave at 93C. The overall heat...
-
American Demographics (Jan. 2002) reported the results of a survey on the planning habits of men and women. In response to the question What is your preferred method of planning and keeping track of...
-
Discuss under what conditions unemployment compensation can be denied to a claimant.
-
Three different plans for financing a $60,000,000 corporation are under consideration by its organizers. Under each of the following plans, the securities will be issued at their par or face amount,...
-
Problem 6. (a) How many quadratic residues are in the residue set Z19? (b) Determine if 4 and 9 are quadratic residues mod 19. (c) Can you solve the congruence x 8 (mod 19)? [6 points]
-
Find the value of K b for the conjugate base of the following organic acids. (a) Picric acid used in the manufacture of explosives; K a = 0.16 (b) Trichloroacetic acid used in the treatment of warts;...
-
Write the chemical equation and use the data in Tables 15.6 and 15.8 to calculate the acid ionization constant for the following ions. (a) Hydroxylammonium ion (b) Ammonium ion Table 15.6 Table 15.8
-
Correct the definition of the italicized term without reference to the text, if correction is needed, so that it is in a form acceptable for publication. Let p be a prime. A p-group is a group with...
-
Pria's friends love to gossip, and they wish Pria would gossip more than she currently does. According to operant conditioning, which scenario is likely to increase Pria's gossiping?
-
The tank shown is filled with water of density p = 1,000 kg/m. What is the hydrostatic pressure force on the slanted surface (1 m by 2.31 m), approximately? 60 3 m 1 m m 2.31 m
-
You wish to buy a home in the stylish Rockhill Gardens section of Waldo. You will need to borrow $289,000 from the bank at 4% interest for 30 years. What will you payment to the bank be? (This amount...
-
4. A firm's most recent dividend was $2.00 and the most recent earnings were $10.00. The firm is expected to grow at 12% for the next 5 years, and then grow forever at 8%. The required rate of return...
-
It is so nice to hear that you have daily communication. I am sure the parents are so thankful for finding out daily what has been done and what is expected at home. Most of my teachers do weekly but...
-
What are the major procedures an audit firm can implement to help ensure that audits are conducted in accordance with due professional care?
-
Prove the formula for (d/dx)(cos-1x) by the same method as for (d/dx)(sin-1x).
-
Given reg [3:0] A = 4'b1101; reg [2:0] B = 3'b111; reg signed [3:0] C = 4'b1101; reg signed [2:0] D = 3'b111; reg signed [7:0] S; Evaluate i. S = A + B; ii. S = A + B + 0; iii. S = C + D; iv. S = C +...
-
Given integer A = 8'shA5; integer B = 8'shB6; integer C = 8'hA5; integer D = 8'hB6; reg signed [31:0] S; Evaluate i. S = A + B; ii. S = A + B + 0; iii. S = C + D; iv. S = C + D + 0; v. S = A + D; vi....
-
A 4-bit magnitude comparator chip (e.g., 74LS85) compares two 4-bit numbers A and B and produces outputs to indicate whether A < B, A = B, or A > B. There are three output signals to indicate each of...
-
Davidson Inc. purchased a machine for $20,000 at the beginning of 2019. It will be depreciated over 5 years on the straight-line basis, with no residual value. For the purpose of this question,...
-
You check to make sure that all the revenue recorded in January was earned during the month. You ask Sally about the Butter and Beans installation work. She says that she has completed about half the...
-
A company that sells its product in a foreign market below the cost of production may be accused of what?

Study smarter with the SolutionInn App


