(a) Determine the charge of the aluminosilicate ion whose composition is AlSi 3 O 10 . (b)...
Question:
(a) Determine the charge of the aluminosilicate ion whose composition is AlSi3O10.
(b) Using Figure 22.32, propose a reasonable description of the structure of this aluminosilicate.
Figure 22.32
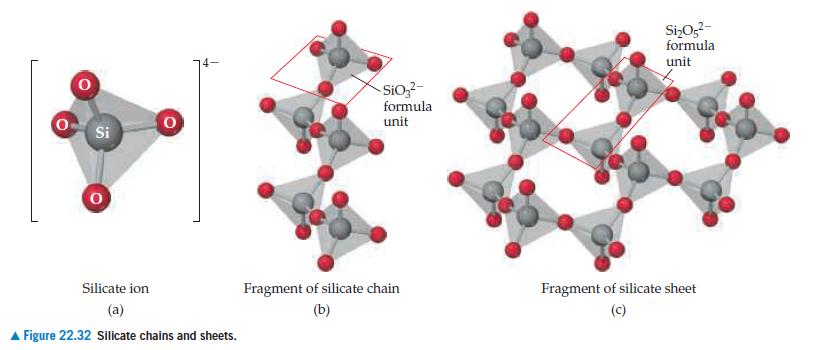
Transcribed Image Text:
Si 0 Silicate ion (a) ▲ Figure 22.32 Silicate chains and sheets. SiO3²- formula unit Fragment of silicate chain (b) Si₂05²- formula unit Fragment of silicate sheet (c)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (6 reviews)
a The charge of the aluminosilicate ion whose composition is AlSi3O10 is 3 b The structure o...View the full answer

Answered By

SHINKI JALHOTRA
I have worked with other sites like Course Hero as a tutor and I have great knowledge on IT skills.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry The Central Science
ISBN: 978-0134414232
14th Edition
Authors: Theodore Brown, H. LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward, Matthew Stoltzfus
Question Posted:
Students also viewed these Sciences questions
-
Determine the charge of the aluminosilicate ion whose composition is AlSi 3 O 10 .
-
Using Table 1.3, determine what charge the ion will carry when each of the following elements reacts to form an ionic compound: Al, Li, S, and O. Table 1.3 Table 1.3 Valence Electrons of the First 18...
-
Propose a reasonable structure for each of the following molecules on the basis of the given molecular formula and of the 1H and proton-decoupled l3C NMR data (a) C7H16O, spectra J and K (* = CH2 by...
-
Is the State of Michigan justified in advertising the prize amount as $1.586 billion? Explain.
-
Jurisdiction E spends approximately $7 million each winter on snow removal. The jurisdiction is considering adding a new income tax provision that would allow people to deduct the cost of snow...
-
In the past few years, the traffic problems in Lynn McKells hometown have gotten worse. Now, Broad Street is congested about half the time. The normal travel time to work for Lynn is only 15 minutes...
-
The adjusted trial balance for Sparkys Electrical Supply, Inc., as of November 30, 2010, is presented next: Requirements 1. Prepare the multi-step income statement for November for Sparkys Electrical...
-
How long does it take for the following to happen? a. $856 grows into $1,122 at 7% b. $450 grows into $725.50 at 12% compounded monthly c. $5,000 grows into $6,724.44 at 10% compounded quarterly
-
Consider how Burlington Ski Lodge could use capital budgeting to decide whether a $15,000,000 lodge expansion would be a good investment. Assume Burlington Ski Lodge's managers developed the...
-
Hotel XYZ is an independent city centre hotel property with 200 rooms. In general its Average Daily rate is approximately 100 throughout the year, and its runs at an average of 65% occupancy. Its...
-
An Iron corrodes to produce rust, Fe 2 O 3 , but other corrosion products that can form are Fe(O)(OH), iron oxyhydroxide, and magnetite, Fe 3 O 4 . (a) What is the oxidation number of Fe in iron...
-
Which of the following statements are true? (a) Si can form an ion with six fluorine atoms, SiF 6 2- , whereas carbon cannot. (b) Si can form three stable compounds containing two Si atoms each, Si 2...
-
The following transactions of Dunn Miles occurred during 2012: Apr 30 Miles is party to a patent infringement lawsuit of $230,000. Miless attorney is certain it is remote that Miles will lose this...
-
This question relates to desired saving, and is based on the table in Question 3 . a. Compute desired saving at each level of disposable income. Plot the saving function on a scale diagram. What is...
-
Consider the following data for a hypothetical economy that produces two goods, milk and honey. a. Compute nominal GDP for each year in this economy. b. Using year 1 as the base year, compute real...
-
For each of the following events, describe the likely effect on real GDP in Canada. a. A major ice storm in Quebec damages homes and thus increases the demand for building materials. b. A flood on...
-
The following diagrams show the \(A D\) and \(A S\) curves in two different economies. a. Explain what aspect of firms' behaviour might give rise to the horizontal \(A S\) curve in Economy \(A\). b....
-
Consider a flow that is made up of straight streamlines emanating from a central point \(\mathrm{O}\), and where the flow velocity along each of the streamlines varies inversely with distance from...
-
Given that Af = af and Ag = bg, where f and g are functions and a and b are constants, under what condition(s) is the linear combination c1f + c2g an eigenfunction of the linear operator A?
-
Bonus shares can be issued out of revenue reserves. True/False?
-
(a) Based on the lattice energies of MgCl2 and SrCl2 given in Table 8.2, what is the range of values that you would expect for the lattice energy of CaCl2? (b) Using data from Appendix C, Figure 7.9,...
-
(a) What is meant by the term covalent bond? (b) Give three examples of covalent bonding. (c) A substance XY, formed from two different elements, boils at -33oC. Is XY likely to be a covalent or an...
-
Which of these elements are unlikely to form covalent bonds: S, H, K, Ar, Si? Explain your choices.
-
Find the slope of the line through the points (-3, 7) and (-9,2). Enter your answer as a simplified improper fraction, if necessary. Do not include "m="in your answer.
-
What is the maximum number of linearly independent vectors that can be found in the nullspace of: A = 120 3 1 24-15 4 36-1 85 4 8 1 12 8
-
The following account balances are taken from the ledger of Maxwell Limited on 31 December 2018, the end of its fiscal year: Maxwell Limited Trial Balance As on 31 December 2018 S'm S'm Investment...

Study smarter with the SolutionInn App


