A laser pointer used in a lecture hall emits light at 650 nm. What is the frequency
Question:
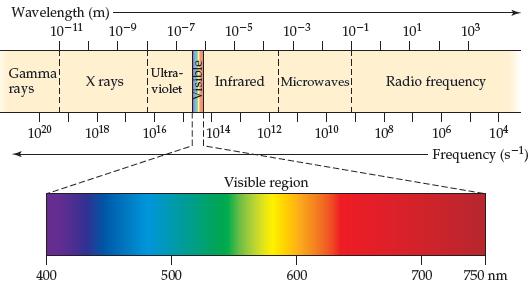
A laser pointer used in a lecture hall emits light at 650 nm. What is the frequency of this radiation? Using Figure 6.4, predict the color associated with this wavelength.
Wavelength (m)- 10-9 10-11 10-7 10-5 10-3 10-1 101 103 Gamma! X rays !Ultra- i violet rays Infrared iMicrowavesi Radio frequency 1020 1018 1016 1014 1012 1010 108 106 104 Frequency (s-1) Visible region 400 500 600 700 750 nm
Step by Step Answer:
The wavelength of a laser is 650 nm 650 10 ...View the full answer



Chemistry The Central Science
ISBN: 9780321910417
13th Edition
Authors: Theodore E. Brown, H. Eugene LeMay, Bruce E. Bursten, Catherine Murphy, Patrick Woodward, Matthew E. Stoltzfus
Related Video
Using a few basic physics principles, you can impress your friends with a trick that makes a bottle disappear. For this experiment, you will need a mini plastic bottle, glycerin, a glass, and a funnel. First, open the bottle and pour some glycerin into it, then close the bottle tightly. Next, pour some water into the glass using a funnel. Place the mini bottle into the glass of water and it will look normal. This is because light travels through air faster than it travels through the glass and water, allowing our eyes to see the bottle inside the glass. However, when you fill the mini bottle with more glycerin, and pour glycerin into the glass, then put the glycerin filled bottle into the glass with glycerin in it. Half of the bottle that is submerged in the glycerin will become invisible as the light travels through glass and glycerin at the same speed, thus it does not bend and no refraction takes place, making the bottle invisible. This happens because both glass and glycerin have almost the same refractive index, which causes the speed of light to be the same in both mediums, causing no bending of light and making the bottle disappear.
Students also viewed these Sciences questions
-
(a) A red laser pointer emits light with a wavelength of 650 nm. What is the frequency of this light? (b) What is the energy of one of these photons? (c) The laser pointer emits light because...
-
(a) A green laser pointer emits light with a wavelength of 532 nm. What is the frequency of this light? (b) What is theenergy of one of these photons? (c) The laser pointer emits light because...
-
A laser used to read CDs emits red light of wavelength 700 nm. How many photons does it emit each second if its power is? (a) 0.10 W, (b) LOW?
-
Determine the force in member GC of the truss and state if this member is in tension or compression. Units Used: kip = 103 lb Given: F1 = 1000 lb F2 = 1000 lb F3 = 1000 lb F4 = 1000 lb a = 10 ft =...
-
y varies jointly as x and z and inversely as the square of w, and y = 12 / 5 when x = 16, z = 3, and w = 5 Find an equation of variation for the given situation.
-
When dealing with the loss of staff, what strategies can be employed?
-
It is estimated that nearly half of all users of the social networking platform TikTok in the United States were between the ages of 10 and 29 in 2020 (Statista.com website). Suppose that the results...
-
(Multiple Choice) 1. All America Bank, the nationwide banking company, owns many types of investments. Assume that All America Bank paid $650,000 for trading securities on December 5. Two weeks...
-
When I am looking for an experimental Modulus of elasticity in a reinforced concrete Beam, where do I start my .1% offset on my load over the deformation graph? Is it .001 or .1% of the total...
-
Refer to Exercise 15.14. Suppose the following input prices are provided for each year: Required: 1. Compute the profit-linked productivity measure. By how much did profits increase due to...
-
The energy from radiation can be used to cause the rupture of chemical bonds. A minimum energy of 242 kJ/mol is required to break the chlorinechlorine bond in Cl 2 . What is the longest wavelength of...
-
(a) Consider the following three statements: (i) A hydrogen atom in the n = 3 state can emit light at only two specific wavelengths, (ii) A hydrogen atom in the n = 2 state is at a lower energy than...
-
True or false: There is no TTL bilateral switch.
-
What are the five different ways to organize your speech? Which one do you plan to use for your next speech and why? Watch the video on YouTube - "Organizational Patterns" and summarize the tips...
-
We asked students of secondary education to provide their ideas on "Online Learning and Teaching" and present them in a document in a format of their preference. In the "Activities" folder you can...
-
The following information about the weekly payroll was obtained from the records of Boltz Co . : Salaries: Deductions: Sales salaries $ 3 2 5 , 0 0 0 Income tax withheld $ 1 1 9 , 0 0 0 Warehouse...
-
On this task you have to evaluate activities and create your own rubric. We asked students of secondary education to provide their ideas on "Online Learning and Teaching" and present them in a...
-
A typical high - income household in 1 9 9 0 earned $ 1 9 1 , 0 2 8 . A similar household in 2 0 1 9 earned $ 2 8 6 , 1 3 8 . What was the relative increase in income for these households from 1 9 9...
-
Talisman Energy and Suncor Energy reported the following investor-related information recently: _____________________ Talisman Energy __________Suncor Energy Earnings per share .............. $1.35...
-
B.) What is the approximate concentration of free Zn 2+ ion at equilibrium when 1.0010 -2 mol zinc nitrate is added to 1.00 L of a solution that is 1.080 M in OH - . For [Zn(OH) 4 ] 2- , K f = 4.610...
-
Three solutions are mixed together to form a single solution. One contains 0.2 mol Pb(CH3COO)2, the second contains 0.1 mol Na2S, and the third contains 0.1 mol CaCl2. (a) Write the net ionic...
-
Which of the following solutions has the largest concentration of solvated protons: (a) 0.2 M LiOH (b) 0.2 M HI (c) 1.0 M methyl alcohol (CH3OH)? Explain
-
Use the molecular representations shown here to classify each compound as either a nonelectrolyte, a weak electrolyte, or a strong electrolyte (see inside back cover for element color scheme). (a)...
-
Multiplication of signed operands, which generate a double-length product in the 2's-complement 100 11 MULTIPLICAND (-13) 0 10 1 1 MULTIPLIER (+11) number system. The general strategy is the...
-
panel = Drawing Panel (160, 160) for i in range (0, 10): panel.draw rectangle (20, 20 + 100 10 * 1, 10 i, 10) Write variations of the above program that draw the figures at the lower right as output....
-
John estimated the following cash flows (in $) for a project: A B 1 Year Cash flow 2 0 -5,700 3 1 1,325 4 2 2,148 5 3 3,528 The required return for the project is 8%. What is the IRR for the project?

Study smarter with the SolutionInn App


