Are these two compounds isomers? Explain. CH-CHCI H-CH3 CH3-CH2-CH-CH2CI
Question:
Are these two compounds isomers? Explain.
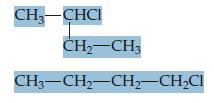
Transcribed Image Text:
CH-CHCI ČH-CH3 CH3-CH2-CH-CH2CI
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
The two compound ha...View the full answer

Answered By

Nimlord Kingori
2023 is my 7th year in academic writing, I have grown to be that tutor who will help raise your grade and better your GPA. At a fraction of the cost on other sites, I will work on your assignment by taking it as mine. I give it all the attention it deserves and ensures you get the grade that I promise. I am well versed in business-related subjects, information technology, Nursing, history, poetry, and statistics. Some software's that I have access to are SPSS and NVIVO. I kindly encourage you to try me; I may be all that you have been seeking, thank you.
4.90+
360+ Reviews
1070+ Question Solved
Related Book For 

Chemistry The Central Science
ISBN: 9780321910417
13th Edition
Authors: Theodore E. Brown, H. Eugene LeMay, Bruce E. Bursten, Catherine Murphy, Patrick Woodward, Matthew E. Stoltzfus
Question Posted:
Students also viewed these Sciences questions
-
Two compounds are isomers if they have the same chemical formula but a different arrangement of atoms. Use Table 8.4 to ÎH estimate for each of the following gas-phase iso-merization reactions,...
-
Two different compounds have the formulation CoBr(SO4) 5NH3. Compound A is dark violet, and compound B is red-violet. When compound A is treated with AgNO3 (aq), no reaction occurs, whereas compound...
-
N-Methylpyrrolidine has a boiling point of 81 °C, and piperidine has a boiling point of 106 °C. (a) Explain this large difference (25 °C) in boiling point for these two isomers. (b)...
-
Use the data from question 13 to produce a cumulative frequency graph and a cumulative relative frequency graph. Question 13 An advertising executive is interested in the age distribution of the...
-
Given the system of equations a1x + b1y = c1, a2x + b2y = c2, Explain why the equations are dependent or the system is inconsistent when
-
Add a new row in the ORDERS table with the following data: Order# = 1021, Customer# = 1009, and Order date = July 20, 2009.
-
Water sloshes back and forth in a tank as shown in Fig. P7.10. The frequency of sloshing, \(\omega\), is assumed to be a function of the acceleration of gravity, \(g\), the average depth of the...
-
The following data are for four independent process-costing departments. Inputs are added continuously. Required: Compute the equivalent units of production for each of the preceding departments...
-
Modern gas turbine engines operate at fuel-to-air ratios of about 0.02, pressure ratios (ratio of compressor exit pressure to inlet pressure) of nearly 40, and with a turbine inlet temperature...
-
Greenscape Pty Ltd provides commercial landscaping services. Linda Drake, the firms owner, wants to develop cost estimates that sh. can use to prepare bids on jobs. After analysing the firm's costs....
-
The liquid substances mercury (density = 13.6 g/mL), water (1.00 g/mL), and cyclohexane (0.778 g/mL) do not form a solution when mixed but separate in distinct layers. Sketch how the liquids would...
-
In what part of the atom does the strong nuclear force operate?
-
Explain why the Harvard Nurses Health Study is an observational study. Critics sometimes say that the results would be more valid if obtained by experiments rather than observations. Discuss whether...
-
(CWE budgeting, 20) The engineer at Clean Water Engineering (CWE) has established a capital investment limit of $1 million for next year's projects that target improved recovery of highly brackish...
-
What does this entry accomplish: debit capital; credit withdrawals?
-
Question 1 An industrial cold room has four 200 mm thick walls made of concrete. The walls are insulated on the outside with a layer of foam 60 mm thick. Cladding with a thickness of 15 mm protects...
-
Harold is completing his travel voucher following a work trip. All purchases greater than $75 require a receipt. Taxi to airport: $27.30 Checked baggage fee: $25.00 Airline: $383.85 Rental Car:...
-
Political Action Committees (PACs) 2023 - 2024 $420,450 Total PAC Money for 2023 - 2024 Number of Contributions: 177 Sectors 2023 - 2024 Sector Total Agribusiness $16,300 Communications/Electronics...
-
Melanie is employed full-time as an accountant for a national hardware chain. She recently started a private consulting practice, which provides tax advice and financial planning to the general...
-
Several months have passed and the Managing Partner approved and properly filed the Complaint and properly submitted the Request for Production of Documents that you drafted. In fact, it has been 75...
-
Using your knowledge of metric units, English units, and the information on the back inside cover, write down the conversion factors needed to convert (a) Mm to nm, (b) Mg to kg, (c) Km to ft (d)...
-
Using your knowledge of metric units, English units, and the information on the back inside cover, write down the conversion factors needed to convert (a) m to mm (b) Ms to ns, (c) Mi to km (d) To L.
-
(a) A bumblebee flies with a ground speed of 15.2 m/s. Calculate its speed in km/h. (b) The lung capacity of the blue whale is 5.0 103 L. Convert this volume into gallons. (c) The Statue of Liberty...
-
List 3 ways to organize a resume and describe when each resume is appropriate
-
Consider the following data for two products of Vigano Manufacturing. Activity Machine setup Parts handling Quality inspections Total budgeted overhead Unit Information Units produced Budgeted Cost...
-
ces Direct Materials Conversion Percent Percent Units Complete Complete Beginning work in process 42,500 inventory Units started this period 165,000 Units completed and 175,000 transferred out 32,500...

Study smarter with the SolutionInn App


