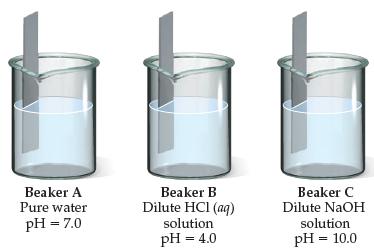
Bars of iron are put into each of the three beakers as shown here. In which beakerA,
Question:
Bars of iron are put into each of the three beakers as shown here. In which beaker—A, B, or C—would you expect the iron to show the most corrosion?
Transcribed Image Text:
Beaker A Pure water Beaker B Dilute HCI (aq) solution Beaker C Dilute NaOH pH = 7.0 solution pH = 4.0 pH = 10.0
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)

Answered By

Marvine Ekina
Marvine Ekina
Dedicated and experienced Academic Tutor with a proven track record for helping students to improve their academic performance. Adept at evaluating students and creating learning plans based on their strengths and weaknesses. Bringing forth a devotion to education and helping others to achieve their academic and life goals.
PERSONAL INFORMATION
Address: , ,
Nationality:
Driving License:
Hobbies: reading
SKILLS
????? Problem Solving Skills
????? Predictive Modeling
????? Customer Service Skills
????? Creative Problem Solving Skills
????? Strong Analytical Skills
????? Project Management Skills
????? Multitasking Skills
????? Leadership Skills
????? Curriculum Development
????? Excellent Communication Skills
????? SAT Prep
????? Knowledge of Educational Philosophies
????? Informal and Formal Assessments
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry The Central Science
ISBN: 9780321910417
13th Edition
Authors: Theodore E. Brown, H. Eugene LeMay, Bruce E. Bursten, Catherine Murphy, Patrick Woodward, Matthew E. Stoltzfus
Question Posted:
Students also viewed these Sciences questions
-
The Evolution of the Fashion Industry: Consider the market for clothes and suppose there exist 100 different styles that can be produced and can be arranged (and equally spaced) on a circle. Among...
-
In the U.S., Presidential elections are not won by the candidate who wins the popular vote nationally. (If they were won in this way, Al Gore would have become President in 2000.) Rather, each state...
-
In cases 1-3 below, assume that Division A has a product that can be sold either to Division B of the same company or to outside customers. The managers of both divisions are evaluated based on their...
-
What is GST?
-
What is meant by indirect least squares? When is it used?
-
Suppose now that the condominium purchasers were all inner-ring people, but that each condominium was constructed from two apartments. What would happen to the price of apartments?
-
How may bank output be measured?
-
1. How might population trends affect a professional services organization like PwC? What might it have to do to adapt to these trends? 2. What challenges might PwC face in adapting to a more diverse...
-
es Cone Corporation is in the process of preparing its December 31, 2024, balance sheet. There are some questions as to the proper classification of the following items: a. $69,000 in cash restricted...
-
In SCTP, a packet is carrying two DATA chunks, each containing 22 bytes of user data. What is the size of each DATA chunk? What is the total size of the packet?
-
The electrodes in a silver oxide battery are silver oxide (Ag 2 O) and zinc. Which electrode acts as the anode?
-
In each of the following balanced oxidationreduction equations, identify those elements that undergo changes in oxidation number and indicate the magnitude of the change in each case. 2MnO 4 - (aq) +...
-
The 2-in.-diameter bar is fixed connected at its ends and supports the axial load P. If the material is elastic perfectly plastic as shown by the stressstrain diagram, determine the smallest load P...
-
A CTOD test is performed on a three point bend specimen. Figure below shows the deformed specimen after it has been unloaded. That is, the displacements shown are the plastic components. P (a) Derive...
-
(2). A minimization LP is being solved by the big M method. e is the excess variable in constraint 1, s2 is the slack variable in constraint 2, and a, a3 are the artificial variables of constraints...
-
Write a program called memo that wraps commands that are to be executed, recording the command line arguments into a memo.history file. The wrapped command should be executed using system() . Ensure...
-
In autarky, country Here produced computers 500,000 computers and 750,000 pairs of shoe each year and country There produces 1,200,000 computers and 600,000 pairs of shoes each year. All the...
-
1/3 + + + + + A B D E The points A, B, C,..., G, and H are equally spaced along the rational number line to the right. Complete parts (a) through (d) + below. (a) What rational number corresponds to...
-
Santana Rey receives the March bank statement for Business Solutions on April 11, 2018. The March 31 bank statement shows an ending cash balance of $67,566. A comparison of the bank statement with...
-
What is a manufacturing system?
-
Chlorine is widely used to purify municipal water supplies and to treat swimming pool waters. Suppose that the volume of a particular sample of Cl2 gas is 8.70 L at 895 torr and 24oC. (a) How many...
-
Many gases are shipped in high-pressure containers. Consider a steel tank whose volume is 55.0 gallons that contains O2 gas at a pressure of 16,500 kPa at 23oC. (a) What mass of O2 does the tank...
-
In an experiment reported in the scientific literature, male cockroaches were made to run at different speeds on a miniature treadmill while their oxygen consumption was measured. In one hour the...
-
ow do synergistic dynamics emerge within high-performance teams, and what role do diverse skill sets, complementary roles, and shared goals play in fostering collaborative innovation and collective...
-
Pietro Frozen Foods, Inc., produces frozen pizzas. For next year, Pietro predicts that 50,000 units will be produced, with the following total costs: Direct materials Direct labor Variable overhead...
-
You will complete two case studies over the course of the semester. These are to be completed individually. Each case study is found in the textbook and there are questions there to help guide your...

Study smarter with the SolutionInn App


