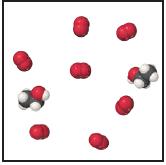
Consider the mixture of ethanol, C 2 H 5 OH, and O 2 shown in the accompanying
Question:
Consider the mixture of ethanol, C2H5OH, and O2 shown in the accompanying diagram.
(a) Write a balanced equation for the combustion reaction that occurs between ethanol and oxygen.
(b) Which reactant is the limiting reactant?
(c) How many molecules of CO2, H2O, C2H5OH, and O2 will be present if the reaction goes to completion?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 90% (10 reviews)
a The given combustion reaction between ethanol and oxygen C 2 H 5 OHl O 2 g CO 2 g H 2 Og Let us ba...View the full answer

Answered By

Nyron Beeput
I am an active educator and professional tutor with substantial experience in Biology and General Science. The past two years I have been tutoring online intensively with high school and college students. I have been teaching for four years and this experience has helped me to hone skills such as patience, dedication and flexibility. I work at the pace of my students and ensure that they understand.
My method of using real life examples that my students can relate to has helped them grasp concepts more readily. I also help students learn how to apply their knowledge and they appreciate that very much.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry The Central Science
ISBN: 9780321910417
13th Edition
Authors: Theodore E. Brown, H. Eugene LeMay, Bruce E. Bursten, Catherine Murphy, Patrick Woodward, Matthew E. Stoltzfus
Question Posted:
Students also viewed these Sciences questions
-
Ethyl alcohol (ethanol) can be produced by the fermentation of sugars derived from trains and other agricultural products. Some countries without large petroleum and natural as reservessuch as...
-
Ethyl alcohol can be bacterially oxidized to acetic acid in the following two-step fermentation sequence: 2 C2H5OH + O2 2 CH3CHO + 2 H2O 2 CH3CHO + O2 2 CH3COOH If the alcohol-containing feedstock...
-
Nitrogen monoxide and oxygen react to form nitrogen dioxide. Consider the mixture of NO and O2 shown in the accompanying diagram. The blue spheres represent N, and the red ones represent O. (a) Draw...
-
In this photo, Ramu is eating raw meat. What do Ramus behaviors suggest about how we develop self-concepts? Would you define Ramu as a human or a wolf?
-
Solve the system of equations using Gaussian elimination or Gauss-Jordan elimination. [6.3] a. x + 2y = 5, 2x - 5y = -8 b. 3x + 4y + 2z = 3, 5x - 2y - 13z = 3, 4x + 3y - 3z = 6
-
For the following exercises, you will address problems and answer questions from management at TAL Distributors. You do not use the TAL Distributors database for any of these exercises. 1. While...
-
Water flows through the orifice meter shown in Fig. P8.122 at a rate of \(0.10 \mathrm{cfs}\). If \(h=3.8 \mathrm{ft}\), determine the value of \(d\). Fig. P8.122 0- 2 in. h
-
During company estimates that total factory overhead costs will be $750,000 for the year. Direct labor hours are estimated to be 300,000. For Darling company, (a) Determine the predetermined factory...
-
Perform an economic analysis studying impacts of Covid-19 on health or health care. For the research bibliography, we need to provide ten (or more) complete research citations. Majority of citations...
-
The Aqua Drilling Company made a contract to drill a well for the Atlas Construction Company. It was expected that this would supply water for a home being constructed by Atlas. Aqua did not make any...
-
If Avogadros number of pennies is divided equally among the 314 million men, women, and children in the United States, how many dollars would each receive? How does this compare with the gross...
-
Consider the mixture of propane, C 3 H 8 , and O 2 shown below. (a) Write a balanced equation for the combustion reaction that occurs between propane and oxygen. (b) Which reactant is the limiting...
-
Draw a diagram depicting the relationship between the general ledger master file, control accounts, subsidiary files, and financial statements.
-
Your company has bonds that mature in 10 years and have a face value of $1,000. The bonds have an 8 percent quarterly coupon (that is, the nominal coupon rate is 8 percent and hence pays $20 every...
-
Simon Company's year-end balance sheets follow. At December 31 Assets Cash Accounts receivable, net Merchandise inventory Prepaid expenses Plant assets, net Total assets Liabilities and Equity...
-
Lisa is a graduate student from Holmes Institute who is actively involved in investment in the securities market. She had established one investment portfolio 5 years ago. Required: Lisa's portfolio...
-
Calculate A. Modify EBIT for cash flow purposes to equal pretax income + interest expense. Then set operating cash flow equal to modified EBIT unlevered taxes + depreciation & amortization. a....
-
ABC Company acquired an asset by way of a six year term finance lease on 1* January 2016. The asset had a fair value of $102,500 and a useful life of five years and the lease contract required 6...
-
On June 5, 2014, Leo purchased and placed in service a new car that cost $20,000. The business use percentage for the car is always 100%. Leo claims any available additional first-year depreciation....
-
Highland Theatre is owned by Finnean Ferguson. At June 30, 2014, the ledger showed the following: Cash, $6,000; Land, $100,000; Buildings, $80,000; Equipment, $25,000; Accounts Payable, $5,000;...
-
(a) What is a hydrocarbon? (b) Butane is the alkane with a chain of four carbon atoms. Write a structural formula for this compound and determine its molecular and empirical formulas.
-
(a) What ending is used for the names of alkanes? (b) Hexane is an alkane whose structural formula has all its carbon atoms in a straight chain. Draw the structural formula for this compound and...
-
(a) What is a functional group? (b) What functional group characterizes an alcohol? (c) With reference to Exercise 2.75, write a structural formula for 1-butanol, the alcohol derived from butane, by...
-
16. Let $V=\operatorname [span]\left\{\left(\begin{array}{1}1 \\ 0 \\ 1 \\ 0\end{array} ight), \left(\begin{array} (1)1 \\ 1 \\ 0 \\ 1\end{array} ight), \left(\begin{array} {1}0 \\ 1 \\ 1 AV...
-
Blossom Ltd., which follows ASPE had the following comparative statement of financial position: Blossom Ltd. Comparative Statement of Financial Position December 31 Assets 2024 2023 Cash $84.000...
-
A survey of the people living in a particular suburb found that the probability that a person above the age of 21 has ever been overseas is $0.27$, and the probability that someone who is 21 years...

Study smarter with the SolutionInn App


