For the molecules shown in 24.85, Data from 24.85 Identify each of the functional groups in these
Question:
For the molecules shown in 24.85,
Data from 24.85
Identify each of the functional groups in these molecules:
(a)
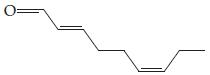
(Responsible for the odor of cucumbers)
(b)
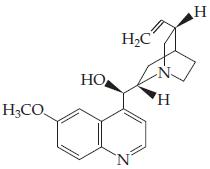
(Quinine — an antimalarial drug)
(c)
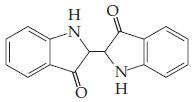
(Indigo — a blue dye)
(d)
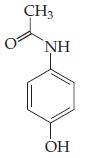
(Acetaminophen — aka Tylenol)
(a) Which one(s) of them, if any, would produce a basic solution if dissolved in water?
(b) Which one(s) of them, if any, would produce an acidic solution if dissolved in water?
(c) Which of them is the most water-soluble?
H2C НО H, H;CO 'N'
Step by Step Answer:
a The compound a has only a carbonyl functional group which is characterized by a carbonoxygen double bond And this functional group does not contain ...View the full answer



Chemistry The Central Science
ISBN: 9780321910417
13th Edition
Authors: Theodore E. Brown, H. Eugene LeMay, Bruce E. Bursten, Catherine Murphy, Patrick Woodward, Matthew E. Stoltzfus
Related Video
Hydrogen peroxide can be used as a mild antiseptic to curb superficial skin infections such as athlete’s foot, but only in diluted quantities. To combat stinky feet, try soaking your feet in a solution of 1 part 3% hydrogen peroxide and 3 parts warm water for 15-20 minutes, then drying them thoroughly. This will kill odor-causing bacteria and soften your feet. To treat athlete\'s foot, you can use a similar solution, but only in diluted quantities, and soak your feet for 30 minutes. Hydrogen peroxide can also be used to keep vegetables fresh by adding 1/4 cup to a bowl of cold water, soaking the vegetables for 20-30 minutes, then draining, drying, and refrigerating them. Alternatively, you can spray vegetables with a solution of 3% hydrogen peroxide and let them stand for a few minutes before rinsing and drying. To keep leftover salad fresh, spray it with a solution of 1/2 cup water and 1 Tbsp. 3% hydrogen peroxide, drain, cover, and refrigerate.
Students also viewed these Sciences questions
-
A simple experiment is set up to measure the effective diffusion coefficient of blue dextran dye into a rigid agarose gel. An aqueous, well-mixed solution of liquid height 10 cm con taining 1.0 g/L...
-
A group of five successful business people were awarded the franchise for one of two new expansion teams in the North American Sports League (NASL). The professional sports franchise was named the...
-
The Stand-Alone Project requires you to perform an in-depth strategic audit of American Airlines, Inc. These past several years have been troubling times for the airline industry. Jet fuel prices...
-
5.Construct a Histogram for the frequency distribution below containing record high temperatures for each of the 50 states. Class boundaries Freq 99.5-104.5 104.5-109.5 109.5-114.5 114.5-119.5...
-
In data involving economic time series such as unemployment, money supply, interest rate, or consumption expenditure, multicollinearity is usually suspected. Why?
-
What does the following SystemVerilog statement do? result = | (data[15:0] & 16'hC820);
-
A SIF uses a single, pneumatic, final element assembly consisting of a 3/2 pilot-operated poppet solenoid valve, a spring-return pneumatic piston actuator, and a floating ball valve. The application...
-
On November 1, 2015, Norwood borrows $200,000 cash from a bank by signing a five-year installment note bearing 8% interest. The note requires equal total payments each year on October 31. Required 1....
-
EEE Co manufactures three products, A, B and C, details of which are as follows: Product A Selling price ($ per unit) 288 Direct materials (kg per unit) 22 Contribution margin ($ per unit) 76 Product...
-
You have observed the following returns over time: Assume that the risk-free rate is 6% and the market risk premium is 5%. a. What are the betas of Stocks X and Y? b. What are the required rates of...
-
(a) Draw the condensed structural formulas for the cis and trans isomers of 2-pentene. (b) Can cyclopentene exhibit cistrans isomerism? Explain. (c) Does 1-pentyne have enantiomers? Explain.
-
Describe what changes occur when atmospheric CO 2 interacts with the world ocean as illustrated here. CO2 (8) CO, (aq) World ean
-
A particle moves along the plane curve C described by r(t) = ti + tj. (a) Find the length of C on the interval 0 t 2. (b) Find the curvature K of the plane curve at t = 0, t = 1, and t = 2. (c)...
-
3 An individual solves an optimal portfolio problem as follows: < E[[ed] W- e-pt 1-7 max E {a(.)} where his wealth evolves according to dW= W.(a:(udt + odZ;) + (1 :)rdt) < Notice that does not appear...
-
After having looked at the different components of emotional intelligence (EI) and assessed your strengths and weaknesses, what insights do you have about yourself? In what ways does your level of EI...
-
The disk in the figure has a mass of m = 0.3 (kg). The initial distance of the disk from the center of rotation is 50 (cm), and the disk slides with a speed of vi = 40(cm/s) The rope was pulled down...
-
The intelligence community has found that the adversary is developing a capability that would be available six years from now. The current U.S. system doesn't have the ability to address the new...
-
All members of the truss shown below have a cross-sectional area of 2 in2 and elastic modulus of 29,000 ksi. 2 k L 4 k (4) 10' (1) 10' (3) (2) Consider the loading at node 4. (a) By hand, use the...
-
The following adjusted trial balance contains the accounts and balances of Cruz Company as of December 31, 2017, the end of its fiscal year. (1) Prepare the December 31, 2017, closing entries for...
-
Two mutually exclusive investment alternatives are being considered. Alternative A requires an initial investment of $20,000 in a machine. Annual operating and maintenance costs are anticipated to be...
-
True or false: (a) The band gap of a semiconductor decreases as the particle size decreases in the 1-10-nm range. (b) The light that is emitted from a semiconductor, upon external stimulation,...
-
What kinds of attractive forces exist between particles in (a) Molecular crystals, (b) Covalent-network crystals, (c) Ionic crystals, (d) Metallic crystals?
-
True or false: If you want a semiconductor that emits blue light, you could either use a material that has a band gap corresponding to the energy of a blue photon or you could use a material that has...
-
Diaz Company issued $91,000 face value of bonds on January 1, Year 1. The bonds had a 9 percent stated rate of interest and a ten- year term. Interest is paid in cash annually, beginning December 31,...
-
Manvir had to make payments of $1,125 every 6 months to settle a $22,000 loan that he received at 4.52% compounded semi-annually. a. How long did it take to settle the loan?
-
If I invest a single amount of $14,000 in an account earning 8% p.a. compounding quarterly for 5 years, how much interest will I have earned in those 5 years?

Study smarter with the SolutionInn App


