Shown here are three pairs of hybrid orbitals, with each set at a characteristic angle. For each
Question:
Shown here are three pairs of hybrid orbitals, with each set at a characteristic angle. For each pair, determine the type of hybridization, if any, that could lead to hybridorbitals at the specified angle.
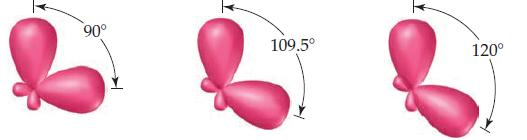
Transcribed Image Text:
90° 109.5° 120°
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 77% (9 reviews)
90 degree angles are formed by d 2 sp 3 orbitals where there is an ...View the full answer

Answered By

Muhammad Ahtsham Shabbir
I am a professional freelance writer with more than 7 years’ experience in academic writing. I have a Bachelor`s Degree in Commerce and Master's Degree in Computer Science. I can provide my services in various subjects.
I have professional excellent skills in Microsoft ® Office packages such as Microsoft ® Word, Microsoft ® Excel, and Microsoft ® PowerPoint. Moreover, I have excellent research skills and outstanding analytical and critical thinking skills; a combination that I apply in every paper I handle.
I am conversant with the various citation styles, among them; APA, MLA, Chicago, Havard, and AMA. I also strive to deliver the best to my clients and in a timely manner.My work is always 100% original. I honestly understand the concern of plagiarism and its consequences. As such, I ensure that I check the assignment for any plagiarism before submission.
4.80+
392+ Reviews
587+ Question Solved
Related Book For 

Chemistry The Central Science
ISBN: 9780321910417
13th Edition
Authors: Theodore E. Brown, H. Eugene LeMay, Bruce E. Bursten, Catherine Murphy, Patrick Woodward, Matthew E. Stoltzfus
Question Posted:
Students also viewed these Sciences questions
-
List causes that could lead to a variable overhead spending variance?
-
List causes that could lead to a variable overhead efficiency variance?
-
List causes that could lead to a fixed overhead spending variance?
-
The distance between the K+ and Cl ions in KCl is 2.80 1010 m. Calculate the energy required to separate the two ions to an infinite distance apart, assuming them to be point charges initially at...
-
Determine whether the ordered pair (1 / 2, 9 / 10) is a solution of the equation 5y - 4 = x.
-
1) you will analyse one of the firm's outstanding bonds. Firstly, calculate the credit spread of this bond, using the yield on Treasury bonds of comparable duration. You will need to compute the rate...
-
Find the indicated z-score(s) shown in the graph. Area = 0.0233 0 z = ? Z
-
The CVP income statements shown below are available for Hoffman Company and Tolan Company. Instructions (a) Compute the degree of operating leverage for each company and interpret your results. (b)...
-
1 Given functions g(x) = and m(z) 2-4, state the domains of the following functions using Interval notation. Domain of 9(2) m(2) Domain of g(m(a)): Domain of m(g(x)) :
-
The following information pertains to Mason Company for Year 2: Beginning inventory .................................90 units @ $40 Units purchased .......................................310 units @...
-
(a) Consider the AF 3 molecules in Exercise 9.27. Which of these will have a nonzero dipole moment? (b) Which of the AF 4 molecules in Exercise 9.28 will have a zero dipole moment?
-
Consider the Lewis structure for glycine, the simplest amino acid: (a) What are the approximate bond angles about each of the two carbon atoms, and what are the hybridizations of the orbitals on each...
-
Hydrogen is the most abundant element in all stars. However, neither absorption nor emission lines due to neutral hydrogen are found in the spectra of stars with effective temperatures higher than 25...
-
Describe the key elements of a theory for qualitative research Explain how the theory supports the proposed study and understanding of the research topic of the gender pay gap for women with...
-
Theory Y management uses the empowerment of workers and gives them the power to report "bad workers" to management so that these bad workers will be reprimanded.
-
Lobster Macaroni and Cheese Find the axis of symmetry, vertex and x-intercepts of the function. Use completing the square to rewrite the function in vertex form. Graph the function. x+2x-3= f(x)
-
Discuss the aircraft inspection criteria used in a comprehensive maintenance program and how the management methods, procedures, and programs are used to prevent bottlenecks in maintenance while...
-
An aluminum shaft of radius R = 30mm is press-fit into a cylindrical hole of a rigid plate. The hole has radius R , where = 0.03mm. The Young modulus of aluminum is 70GPa, and its Poisson ratio is =...
-
Referring to the data presented in E15-12 for Wolstenholme Corp., assume the payments are annual fi xed principal payments instead of blended payments. Instructions (a) Calculate the annual principal...
-
Prepare a stock card using the following information A company is registered for GST which it pays quarterly, assume GST was last paid on the 30th of June 2019. It uses weighted average cost...
-
Imagine a container placed in a tub of water, as depicted in the accompanying diagram. (a) If the contents of the container are the system and heat is able to flow through the container walls, what...
-
The automobile fuel called E85 consists of 85% ethanol and 15% gasoline. E85 can be used in so-called "flex-fuel" vehicles (FFVs), which can use gasoline, ethanol, or a mix as fuels. Assume that...
-
At 20C (approximately room temperature) the average velocity of N2 molecules in air is 1050 mph. (a) What is the average speed in m/s? (b) What is the kinetic energy (in J) of an N2 molecule moving...
-
Hi, could someone please help me with this. I need the letter to be nearly two hundred words. This is the assignment below: You are an architect who is working for Dream Homes Company in Toronto....
-
Discussion Board Post - Read the speech example from the textbook below. Then answer the questions in blood at the bottom. "As a veterinarian and owner of a small-animal practice, you work closely...
-
The Essentials of Technical Communication (Tebeaux & Dragga), Oxford U. Press, Third Edition - I. Please explain how a proposal is an argumentative or persuasive piece of writing -- and the four key...

Study smarter with the SolutionInn App


