Snapshots of two hypothetical reactions, A(g) +B(g) AB(g) and X(g) + Y(g) XY(g) at five
Question:
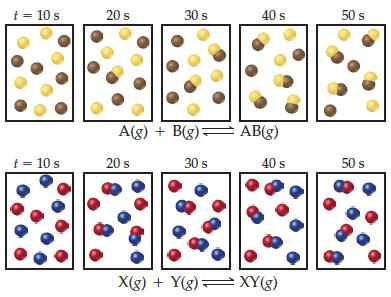
Snapshots of two hypothetical reactions, A(g) +B(g) ⇌ AB(g) and X(g) + Y(g) ⇌ XY(g) at five different times are shown here. Which reaction has a larger equilibrium constant?
Transcribed Image Text:
t = 10 s 20 s 30 s 40 s 50 s A(g) + B(g)= AB(g) t= 10 s 20 s 30 s 40 s 50 s X(g) + Y(g) : = XY(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
Equilibrium constant is mainly dependent on temperature of t...View the full answer

Answered By

Suneel Kumar
I completed my masters in Accountancy and finance and started doing job . I have a good enough interest and expertise in teachin g students. Exploring subject through a tutor will make me a good knowledge.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry The Central Science
ISBN: 9780321910417
13th Edition
Authors: Theodore E. Brown, H. Eugene LeMay, Bruce E. Bursten, Catherine Murphy, Patrick Woodward, Matthew E. Stoltzfus
Question Posted:
Students also viewed these Sciences questions
-
1. A firm consists of 600 acres of land, of which 500 acres will be planted with corn, soybeans, and wheat according to this condition: -At least half of the planted acreage should be in corn - No...
-
24. A uniform chain of length L and mass M is lying on a smooth table and one third of its length is hanging vertically down over the edge of the table. If g is acceleration due to gravity, the work...
-
A culture of yeast that requires uracil for growth (urα3ÍË was mutagenized, and two mutant colonies, X and Y, have been isolated. Mating type a cells of mutant X arc mated...
-
The average of 4 consecutive odd positive integers is 16. The product of the smallest and largest positive odd integer is 1. 210 2. 247 3. 294 4. 320 None of the above 5.
-
Water flows steadily into a well-insulated electrical water heater (see Anim. 4- 1-1) with a mass flow rate of 1 kg/s at 100 kPa and 25C. Determine: The electrical power consumption if the water...
-
Sweeten Company had no jobs in progress at the beginning of March and no beginning inventories. The company has two manufacturing departmentsMolding and Fabrication. It started, completed, and sold...
-
The budgeted monthly service revenues for Shumin Services Ltd for January to June are as follows. All of the firms invoicing for services is on a credit basis. All payments received from clients on...
-
On January 1, 2012, Pedraza Corporation issued $1,000,000 face value, 6%, 10-year bonds at $1,077,217. This price resulted in an effective-interest rate of 5% on the bonds. Pedraza uses the...
-
Mountain Dew Partnership (MDP) was setup 8 years ago. It was formed by 2 brothers Tawanda and Tendai and their best friend Brian. Tawanda and Tendai are originally from Zimbabwe but are now British...
-
Mike?s Custom Furniture, Inc., reported the following comparative income statements for the years ended April 30, 2018, and April 30, 2017. During 2018, Mike?s Custom Furniture, Inc., discovered that...
-
As shown in Figure 14.24, the first step in the heterogeneous hydrogenation of ethylene is adsorption of the ethylene molecule on a metal surface. One proposed explanation for the sticking of...
-
Suppose that, in the absence of a catalyst, a certain biochemical reaction occurs x times per second at normal body temperature (37C). In order to be physiologically useful, the reaction needs to...
-
Debra and Glen are partners who agree that Debra will receive a $100,000 salary allowance after which remaining incomes or losses will be shared equally. If Glens capital account is credited $8,000...
-
Write email to a client, following the Direct method of Delivering Bad News, that explains why you are unable to carry their new line of products. You can be creative with the details, but there...
-
Siyanqoba Trade Union is in negotiations with DD Manufacturers about a wage increase. The parties fail to agree, and the matter is referred to the CCMA for conciliation. After two weeks the matter is...
-
Why do firms expand into other countries and how to adjust the economic/political risk in international capital budgeting? What are prerequisites to be a global financial center?
-
Perform the indicated operations and simplify completely: (x+9) - (x+5) Answer: =
-
Estimate the limit 4 -1 lim- x0 cosx1
-
Use the five-year summary of selected financial data (rounded) for Canadian Tire to answer the following questions. Requirements 1. Using 2010 as the base year, perform trend analysis of Canadian...
-
Which of the followingcarbocations is the least stable? CH3CH2 . CH3CHCH3 CH3 I . CH3C0 T CH3 IV. V. CH3 CH3CCH2 CH3
-
The first ionization energy and electron affinity of Ar are both positive values. (a) What is the significance of the positive value in each case? (b) What are the units of electron affinity?
-
If the electron affinity for an element is a negative number, does it mean that the anion of the element is more stable than the neutral atom? Explain.
-
Although the electron affinity of bromine is a negative quantity, it is positive for Kr. Use the electron configurations of the two elements to explain the difference.
-
Hall & Company issues $ 1 0 0 , 0 0 0 , 0 0 0 in 6 % bonds with detachable warrants of 1 0 warrants for each $ 1 , 0 0 0 bond. The bonds are issued at 9 9 . Record the issuance under the following...
-
Assume that hein acquired Dolan Development last year. Hein recorded the following intangible assets on the date of acquisition: Goodwill: $ 1 , 5 0 0 , 0 0 0 Dolan Development trademark: $ 6 0 0 , 0...
-
What is the total amount of interest that will be paid of the loan is carried to term with no extra payments made building cost 3 3 0 0 0 0 . loan started im Dec 2 0 2 3 worh a 7 . 5 % interest rate...

Study smarter with the SolutionInn App


