The color and wavelength of the absorption maximum for [Ni(H 2 O) 6 ] 2+ , [Ni(NH
Question:
The color and wavelength of the absorption maximum for [Ni(H2O)6]2+, [Ni(NH3)6]2+, and [Ni(en)3]2+ are given in Figure 23.30. The absorption maximum for the [Ni(bipy)3]2+ ion occurs at about 520 nm.
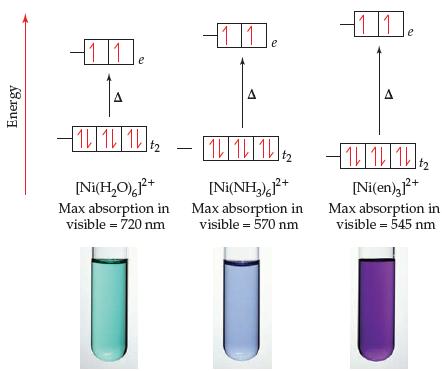
(a) What color would you expect for the [Ni(bipy)3]2+ ion?
(b) Based on these data, where would you put bipy in the spectrochemical series?
Transcribed Image Text:
11 11 11 A IL 1L 12 1 1 1 [Ni(H,O0),F+ Max absorption in visible = 720 nm [Ni(en),+ [Ni(NH,),P* Max absorption in visible = 570 nm Max absorption in visible = 545 nm Energy
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 84% (13 reviews)
a Green and Violet It absorbs 725 nm red light and appears as the complem...View the full answer

Answered By

Madhur Jain
I have 6 years of rich teaching experience in subjects like Mathematics, Accounting, and Entrance Exams preparation. With my experience, I am able to quickly adapt to the student's level of understanding and make the best use of his time.
I focus on teaching concepts along with the applications and what separates me is the connection I create with my students. I am well qualified for working on complex problems and reaching out to the solutions in minimal time. I was also awarded 'The Best Tutor Award' for 2 consecutive years in my previous job.
Hoping to get to work on some really interesting problems here.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry The Central Science
ISBN: 9780321910417
13th Edition
Authors: Theodore E. Brown, H. Eugene LeMay, Bruce E. Bursten, Catherine Murphy, Patrick Woodward, Matthew E. Stoltzfus
Question Posted:
Students also viewed these Sciences questions
-
The [Ni (H2O)6]2+ ion has an absorption maximum at about 725 nm, whereas the [Ni (H2O)6]2+ ion absorbs at about 570nm. Predict the color of a solution of each ion. (b) The [Ni(en)3]2+ ion occurs at...
-
in the Maller-Lyer illusion it is believed that the fin angles act as depth cues. That is from angles 105" to 165" our brains interpret it as an edge coming towards us and angles 15 to 75" appear as...
-
What products would you expect from the reaction of 1 mol of 1,3-butadiene and each of the following reagents? (If no reaction would occur, you should indicate that as well.) (a) 1 mol of Cl2 (b) 2...
-
Given that a quantity Q(t) is described by the exponential growth function Q(t) = 400e0.01t where t is measured in minutes, answer the following questions. (a) What is the growth constant k? k = (b)...
-
Consider Figure 9-10, which plots the gross domestic product (GDP) growth, in percent, against the ratio of investment/GDP, in percent, for several countries for 1974 to 1985.28 The various countries...
-
Write a Boolean equation in sum-of-products canonical form for each of the truth tables in Figure 2.81. (a) (b) (c) (d) (e) A 1 O HOH O HOH
-
What aerodynamic factors affect induced drag?
-
Qen and Associates wants to buy automated coffee roaster/grinder/ brewer. This piece of equipment would have a useful life of six years, would cost $218,500, and would increase annual net inflows by...
-
2. Using the definition of the derivative, use an appropriate value of h to approximate the derivative of f'(2) where f(x) = x* 3. Find the derivative for the following functions: (a) f(x) =...
-
Richard chooses technique 0 and 2 requiring 10+10-20 efforts and provising 10+11=21 benefits. Hence, 21 is returned as the output Example 2: input1: 3 input2: (10,10,10,10) input3: (10,11,12,15)...
-
The colors in the copper-containing minerals malachite (green) and azurite (blue) come from a single d-d transition in each compound. (a) What is the electron configuration of the copper ion in these...
-
Give the number of (valence) d electrons associated with the central metal ion in each of the following complexes: (a) K 3 [Fe(CN) 6 ] (b) [Mn(H 2 O) 6 ](NO 3 ) 2 (c) Na[Ag(CN) 2 ] (d) [Cr(NH 3 ) 4...
-
St. Joseph's Hospital began operations in December 2014 and had patient service revenues totaling $950,000 (based on customary rates) for the month. Of this, $103,000 is billed to patients,...
-
Using the information, calculate the followings. Operating activities Net earings Adjustments to reconcile net eamings to cash provided by (used in) operating activities: Depreciation and...
-
1. "The Greenhouse effect is a natural phenomenon*. Briefly explain 2. Using the greenhouse effect, explain how the burning of fossil fuels contribute to global warming 3. Briefly outline TWO...
-
Suppose the following graph shows the annual domestic market for cheese in Algeria, which is a producer as well as an importer of cheese. The free- trade world price of cheese is P per ton. PRICE...
-
Sinner Service Inc. (SSI) has the following 2, semi-annual coupon bond issues outstanding: Bond Coupon Rate 1 8% 2 7% Price Quote 97.65 102.55 Maturity 5.5 years 10 years Face Value $900,000 $500,000...
-
Make a flow chart on a process you know of. Run Chart and Control Chart: Make a data set based on the performance of your process and prepare a run chart and control chart. Cause and Effect Diagram:...
-
Prepare journal entries to record the following merchandising transactions of Cabela's, which uses the perpetual inventory system and the gross method. July 1 Purchased merchandise from Boden Company...
-
What is the maximum volume of 0.25 M sodium hypochlorite solution (NaOCl, laundry bleach) that can be prepared by dilution of 1.00 L of 0.80 M NaOCl?
-
Although polyethylene can twist and turn in random ways, the most stable form is a linear one with the carbon backbone oriented as shown in the following figure: The solid wedges in the figure...
-
(a) In polyvinyl chloride shown in Table 12.5, which bonds have the lowest average bond enthalpy? (b) When subjected to high pressure and heated polyvinyl chloride converts to diamond. During this...
-
Silicon has the diamond structure (Figure 12.30(a)) with unit cell edge length of 5.43 and eight atoms per unit cell. (a) How many silicon atoms are there in of material? (b) Suppose you dope that...
-
Question 1: How do strategy and tactical action relate to each other in an organization? Question 2: How can you improve the implementation of strategy in this business organization?...
-
Larkin Corporation sells products for $125 each that have variable costs of $50 per unit. Larkin's annual fixed costs are $430,000. (10 points Total) a. What is Larkin's contribution margin per unit?...
-
Find the determinant of the linear transformation T(M): [3] M from the space V of 22 upper triangular matrices to V.

Study smarter with the SolutionInn App


