The three graphs below show the variation in radius, effective nuclear charge, and maximum oxidation state for
Question:
The three graphs below show the variation in radius, effective nuclear charge, and maximum oxidation state for the transition metals of period 4. In each part below identify which property is being plotted.
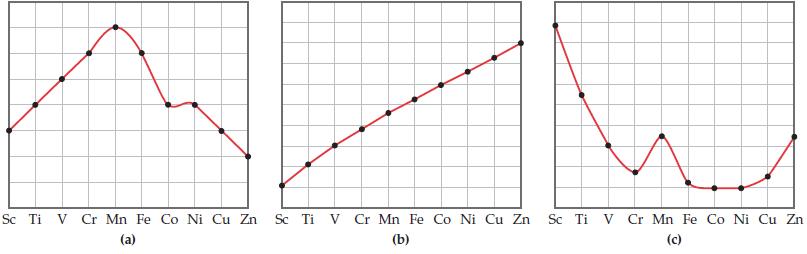
Sc Ti V Cr Mn Fe Co Ni Cu Zn Sc Ti V Cr Mn Fe Co Ni Cu Zn Sc Ti V Cr Mn Fe Co Ni Cu Zn (a) (b) (c)
Step by Step Answer:
a Maximum oxidation state dblock elements exhibit a variety of oxidation states A complete or halffu...View the full answer



Chemistry The Central Science
ISBN: 9780321910417
13th Edition
Authors: Theodore E. Brown, H. Eugene LeMay, Bruce E. Bursten, Catherine Murphy, Patrick Woodward, Matthew E. Stoltzfus
Related Video
Lemon juice preserves apples by slowing down the oxidation process. Oxidation is a chemical reaction that occurs when oxygen reacts with certain substances, such as apples. When an apple is cut or bitten, oxygen is exposed to the inside of the apple and causes enzymes in the apple to turn brown, which is an indication of oxidation. The browning process is caused by the production of polyphenol oxidase (PPO) enzymes that convert phenolic compounds into quinones, which then polymerize to form the brown pigments. One of the compounds present in lemon juice is ascorbic acid (vitamin C), which is a natural antioxidant. Antioxidants work by neutralizing the free radicals that cause oxidation. When lemon juice is applied to apples, the ascorbic acid in the lemon juice reacts with the PPO enzymes and slows down the browning process. You can do an experiment by cutting apples into small pieces, leaving one apple piece in contact with air and the others covered with lemon juice and compare the browning process. This will help to understand the antioxidation process in fruits.
Students also viewed these Sciences questions
-
O n the same graph, plot the effective nuclear charge and atomic radius (see Figure 8.5) versus atomic number for the second period elements Li to Ne. Comment on the trends.
-
Consider the iso-electronic ions F- and Na+. (a) Which ion is smaller? (b) Using Equation 7.1 and assuming that core electrons contribute 1.00 and valence electrons contribute 0.00 to the screening...
-
Consider the iso-electronic ions Cl- and K+. (a) Which ion is smaller? (b) Using Equation 7.1 and assuming that core electrons contribute 1.00 and valence electrons contribute nothing to the...
-
Consider thedeadlock situation that could occur in the dining-philosophers problem when the philosophers obtain the chopsticks one at a time. Discuss how the four necessary conditions for deadlock...
-
Would you expect heteroscedasticity to be present in the following regressions? Sample Net worth Log of net worth (a) Corporate profits Fortune 500 (b) Log of corporate (c) Dow Jones industria (o)...
-
A particular DSL modem operates at 768 kbits/sec. How many bytes can it receive in 1 minute?
-
Explain what type of stability the aircraft would exhibit if the CG is behind the CL.
-
Presto Company makes radios that sell for $30 each. For the coming year, management expects fixed costs to total $220,000 and variable costs to be $18 per unit. (a) Compute the break-even point in...
-
Apex Company prepared the statement of cash flows for the current year that is shown below: Apex Company Statement of Cash Flows-Indirect Method Operating activities: Net income Adjustments to...
-
Richard chooses technique 0 and 2 requiring 10+10-20 efforts and provising 10+11=21 benefits. Hence, 21 is returned as the output Example 2: input1: 3 input2: (10,10,10,10) input3: (10,11,12,15)...
-
Using Gf for ozone from Appendix C, calculate the equilibrium constant for Equation 22.24 at 298.0 K, assuming no electrical input. electricity 3 02(g) 2 03(g) AH 285 k %3D
-
The lanthanide contraction explains which of the following periodic trends? (a) The atomic radii of the transition metals first decrease and then increase when moving horizontally across each period....
-
Which of the following outcomes is most likely a result of globalization? A. Unequal gains B. Increased independence C. Decreased access to talent
-
Write a C++ program to compute the real roots of the equation: ax2+bx+c=0. The program will prompt the user to input the values of a, b, and c. It then computes the real roots of the equation based...
-
What are the implications of distributed leadership models for organizational effectiveness and innovation, as leaders decentralize authority and empower employees at all levels to take ownership of...
-
World Fossil Fuel Regulation At the continued rate of extraction, in how many years is the UK Coal Energy coal from the Folen Mine likely to run out?
-
Identify three milestones or key times throughout the leasing campaign of4/17 Wonderview Boulevard, Fantasy Glen when you would report on the effectiveness of the campaign so far.
-
Both assets depend on the state of the local economy. Three possible developments of the state of the local economy for the next couple of years are relevant. Upsize, downsize and remain. Development...
-
Confucius Bookstore's inventory is destroyed by a fire on September 5, 2017. The following data for year 2017 are available from the accounting records. Estimate the cost of the inventory destroyed....
-
The area of square PQRS is 100 ft2, and A, B, C, and D are the midpoints of the sides. Find the area of square ABCD. B A
-
Referring to Figure 11.28, describe all the phase changes that would occur in each of the following cases: (a) Water vapor originally at 0.005 atm and - 0.5 oC is slowly compressed at constant...
-
The molecules Have the same molecular formula (C3H8O) but different normal boiling points, as shown. Rationalize the difference in boiling points? (b) Ethyl methyl ether (a) Propanol 97.2C 10.8 C
-
Referring to Figure 11.29, describe the phase changes (and the temperatures at which they occur) when CO2 is heated from - 80 oC to - 20 oC at (a) A constant pressure of 3 atm, (b) A constant...
-
Benson and Orton are partners who share income in the ratio of 2:3 and have capital balances of $50,000 and $30,000 respectively. Ramsey is admitted to the partnership and is given a 40% interest by...
-
Compute ending work in process inventory for a manufacturer using the following information. Raw materials purchased Direct materials used Direct labor used Factory overhead Work in process...
-
A spring with spring constant 80 N/m is placed at the bottom of a 6 m high ramp with opening angle 60 . A 3 kg mass is then placed on the spring and pushed down so the spring is compressed by a...

Study smarter with the SolutionInn App


