This figure shows the interaction of a cation with surrounding water molecules. Would you expect the energy
Question:
This figure shows the interaction of a cation with surrounding water molecules.
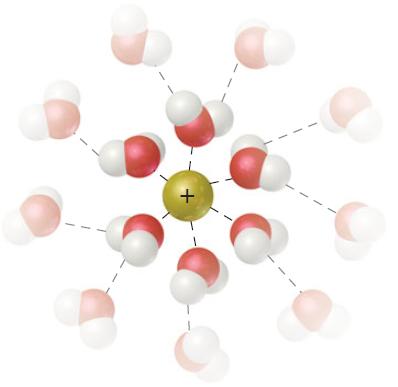
Would you expect the energy of ion–solvent interaction to be greater for Na+ or Li+? Explain.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (12 reviews)
Ionsolvent interactions inversely proportion al to the size of the ion Ie smaller is ...View the full answer

Answered By

Umair Expert
Hi Everyone.
I have 6 years of teaching experience.
I am serving as a tutor 2 more websites.
I will provide you projects and solutions of questions related to any subject.
I am a good programmer.(PHP, Python)
I am good command in mathematics.
I am here to assist you.
I'll be very happy to work with you...
Thanks dear..
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Question Posted:
Students also viewed these Sciences questions
-
Which of the following molecules would you expect to be aromatic? (a) (b) (c) (d) (e) (f) (g) (h) (i) (j) (k) (l) N+
-
Would you expect alanine (an amino acid) to be more soluble in water or in hexane? Explain.
-
The accompanying graph depicts the interaction energy between two water molecules situated so that their dipole moments are parallel and pointing in the same direction. Sketch an approximate curve...
-
The Federal Reserve may raise its benchmark interest rate later this month. How is this achieved? Why would they do this? Explain the consequences fully. (Include graphs with your answer)
-
Find and s for: (a) The lizard data in Exercise 2.19. (b) The acid rain data in Exercise 2.115. 1.28 1.36 1.24 2.47 1.94 2.52 2.67 1.29 1.56 2.66 2.17 57 2.10 2.54 163 2.11 2.57 1.72 0.76 1.02 1.78...
-
Casinos are required to verify that their games operate as advertised. American roulette wheels have 38 slots18 red, 18 black, and 2 green. In one casino, managers record data from a random sample of...
-
A manufacturer determines that a big screen HDTV set had probabilities of \(0.8,0.15,0.05\), respectively, of being placed in the categories acceptable, minor defect, or major defect. If 3 HDTVs are...
-
Kristin Simon needs to borrow $4,000. First State Bank will lend her the money for 12 months through a single-payment loan at 8 percent, discount; Home Savings and Loan will make her a $4,000,...
-
Prepare the four required closing entries for a company that has the following account balances. Capital Stock 500,000 Rent Revenue 5/s 200,000 Fees Earned 100,000 325,000 300,000 Wage Expense...
-
1. (Cost of trade credit) Calculate the effective cost of the following trade credit terms when payment is made on the net due date: 2. (Annual percentage yield) Compute the cost of the trade credit...
-
What molecular structural features cause high-density polyethylene to be denser than low-density polyethylene?
-
List four properties of a solution that depend on the total concentration but not the type of particle or particles present as solute.Write the mathematical expression that describes how each of...
-
In what way might the first-come, first-served rule be unfair to the customer waiting for service in a bank or hospital?
-
People post a lot of pictures on Facebook of themselves and their friends drinking. Do you think that people who spend a lot of time visiting these Facebook pages would cultivate a view of reality...
-
Apply any element of Dramatism to your life. For instance, have you experienced identification with a public figure? If so, what strategies did they employ that allowed you to feel this way?
-
Giles contends that self-perception can influence the accommodation process. How does a persons self-perception affect how one chooses whether or not to accommodate? Provide examples to illustrate...
-
Aristotles critics have focused on the fact that his theory is simply a collection of lecture notes that are contradictory, vague, and often narrow. Does this make a difference to you? Why or why not?
-
We have noted that overaccommodation typically results in miscommunication. Some research in Communication Accommodation Theory has also examined underaccommodation in your college and/or family, or...
-
On March 15, 2015, Helen purchased and placed in service a new Escalade. The purchase price was $62,000, and the vehicle had a rating of 6,500 GVW. The vehicle was used 100% for business. Calculate...
-
Why do bars offer free peanuts?
-
Air in an automobile tire is maintained at a pressure of 220 kPa (gauge) in an environment where the atmospheric pressure is 94 kPa. The air in the tire is at the ambient temperature of 25C. Now a...
-
The thrust developed by the engine of a Boeing 777 is about 380 kN. Assuming choked flow in the nozzle determine the mass flow rate of air through the nozzle. Take the ambient conditions to be 265 K...
-
A stationary temperature probe inserted into a duct where air is flowing at 250 m/s reads 85C. What is the actual temperature of air?
-
1. Solve FV = PMT((1+r/m)mt 1) for PMT. PMT = r/m 2. Given the formula in the format to input into the calculator. PMT =
-
Bramble Enterprises purchased a machine on January 1 , 2 0 2 4 , for $ 2 2 1 0 0 . The machine had an estimated useful life of 1 0 years and an estimated residual value of $ 2 7 0 0 . Assuming...
-
For an ideal gas, what is the pressure in atm if the volume of the container is 5000 mL, the mol quantity is 0.812 mol, and the temperature is 28 degrees Celsius? PV = nRT (qu - A) (BA/MA + d) = nRT

Study smarter with the SolutionInn App


