If possible, find each of the following. (a) A + B (b) 3A (c) 2A - 3B
Question:
If possible, find each of the following.
(a) A + B
(b) 3A
(c) 2A - 3B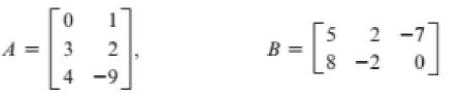
Transcribed Image Text:
A || 1 3 2 4-9 2-7 B= B - [ - -?] 8-2 0
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (6 reviews)
a b c A B is unde...View the full answer

Answered By

ANDREW KIPRUTO
Academic Writing Expert
I have over 7 years of research and application experience. I am trained and licensed to provide expertise in IT information, computer sciences related topics and other units like chemistry, Business, law, biology, biochemistry, and genetics. I'm a network and IT admin with +8 years of experience in all kind of environments.
I can help you in the following areas:
Networking
- Ethernet, Wireless Airmax and 802.11, fiber networks on GPON/GEPON and WDM
- Protocols and IP Services: VLANs, LACP, ACLs, VPNs, OSPF, BGP, RADIUS, PPPoE, DNS, Proxies, SNMP
- Vendors: MikroTik, Ubiquiti, Cisco, Juniper, HP, Dell, DrayTek, SMC, Zyxel, Furukawa Electric, and many more
- Monitoring Systems: PRTG, Zabbix, Whatsup Gold, TheDude, RRDtoo
Always available for new projects! Contact me for any inquiries
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

College Algebra With Modeling And Visualization
ISBN: 9780134418049
6th Edition
Authors: Gary Rockswold
Question Posted:
Students also viewed these Mathematics questions
-
Refer to the periodic table (Figure 2.15 or inside front cover) and obtain the group and period for each of the following elements. Also determine whether the element is a metal, nonmetal, or...
-
Emily Jackson (Social Security number 765-12-4326) and James Stewart (Social Security number 466-74-9932) are partners in a partnership that owns and operates a barber shop. The partnership's first...
-
Clifford Johnson has a limited partnership investment and a rental condominium. Clifford actively manages the rental condominium. During 2019, his share of the loss from the limited partnership was...
-
Which of the following is most nearly the mass of the Earth? (The radius of the Earth is about 6.4 106 m) A. 6 x 1024 kg B. 6 x 107 kg C. 6 x 103 kg D. 6 x 1033 kg E. 6 x 1036 kg
-
1. Distinguish among the owner, the insured, the beneficiary, and the contingent beneficiary of a life insurance policy. 2. Briefly describe each of the five components of all insurance policies. 3....
-
A steel alloy contains 97.5 wt% Fe, 2.0 wt% Mo, and 0.5 wt% C. (a) What is the eutectoid temperature of this alloy? (b) What is the eutectoid composition? (c) What is the proeutectoid phase? Assume...
-
Juliette Shulof Furs (JSF) was a New York corporation that had been in the fur-dealing business for 15 years. George Shulof, an officer of JSF, attended two auctions conducted by Finnish Fur Sales...
-
Jobs, Inc. has recently started the manufacture of Tri-Robo, a three-wheeled robot that can scan a home for fires and gas leaks and then transmit this information to a mobile phone. The cost...
-
The figure depicts Jack - in - the - box: "Jack" is attached inside a box by a spring, as shown. You estimate Jack's mass to be 0 . 4 k g . As so often happens, a sign tells you the spring constant:...
-
Graph the solution set to the inequality. 2x - y <1
-
Solve the equation for x and then solve it for y. x - 2 = 5
-
The monopolist produces at the minimum point of her ATC curve _________-. a) all the time b) most of the time c) some of the time d) none of the time
-
MT can correct speaker's grammar without changing meaning. Question 24 options: True False
-
You have a loan outstanding. It requires making seven annual payments of $5,000 each at the end of the next seven years. Your bank has offered to allow you to skip making the next six payments in...
-
The Parol Evidence Rule prevents parties from introducing evidence of other issues when a contract is seen as representing the entire agreement. There are a few exceptions. One is that the parties...
-
E.12.3 investment in an item of equipment is $22,000. It has a five-year life and no salvage value and shirt one depreciation method is used the equipment is expected to provide an annual savings of...
-
Corporate E&P is $100,000. Cindys tax basis in her shares is $1,000,000 ($5,000 per share). She receives $800,000 in exchange for her 50 redeemed shares. IGNORING the correct answer, would Cindy...
-
A loaded tractor-trailer with a total mass of 5000 kg traveling at hits a loading dock and comes to a stop in 0.64 s. What is the magnitude of the average force exerted on the truck by the dock?
-
A 2500-lbm car moving at 15 mi/h is accelerated at a constant rate of 15 ft/s 2 up to a speed of 50 mi/h. Calculate force and total time required?
-
Consider the regression model fit to the arsenic data in Exercise 12-16. Use arsenic in nails as the response and age, drink use, and cook use as the regressors. (a) Calculate 99% confidence...
-
Consider the regression model fit to the coal and limestone mixture data in Exercise 12-17. Use density as the response. (a) Calculate 90% confidence intervals on each regression coefficient. (b)...
-
Consider the regression model fit to the nisin extraction data in Exercise 12-18. (a) Calculate 95% confidence intervals on each regression coefficient. (b) Calculate a 95% confidence interval on...
-
In ethylene glycol production plant, ethylene oxide is removed from water-glycols mixture in a flash column operating constant temperature of 105C and a constant pressure of 1 atm. At steady state...
-
The system given in the figure is h1-30 cm, h2= 10 cm, h3-70 cm, h4= 20 cm and the settings of the restrictions pi = 997 kg/m3, pj-1.25 kg/m3 "It is not fixed!", pk = 13600 kg/m3. Since the pressure...
-
1. Calculate (a) (b) the pH when 15 mL of 0.25 M HClO4 are added to 25 mL of 0.20 M NaOH. the pH when 25.0 mL of 0.40 M HCl are added to 40.0 mL of 0.30 M NH3. (Dissociation constant for ammonia is...

Study smarter with the SolutionInn App


