In Exercises 140, use properties of logarithms to expand each logarithmic expression as much as possible. Where
Question:
In Exercises 1–40, use properties of logarithms to expand each logarithmic expression as much as possible. Where possible, evaluate logarithmic expressions without using a calculator.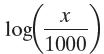
Transcribed Image Text:
log X 1000
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
log X ...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Mathematics questions
-
In Exercises 140, use properties of logarithms to expand each logarithmic expression as much as possible. Where possible, evaluate logarithmic expressions without using a calculator. (125) logs
-
In Exercises 140, use properties of logarithms to expand each logarithmic expression as much as possible. Where possible, evaluate logarithmic expressions without using a calculator. In e2 5
-
In Exercises 140, use properties of logarithms to expand each logarithmic expression as much as possible. Where possible, evaluate logarithmic expressions without using a calculator. logg (13.7)
-
Determine the force in members HI, FI, and EF of the truss, and state if the members are in tension orcompression. 3 m D. -2 m--2 m2 m--2 m--2 m--2 m- 4 kN 5 KN 6 kN 8 kN
-
Womens height is a suspected factor for difficult deliveries, that is, shorter women are more likely to have Caesarean sections. A medical researcher found in a sample of 45 women who had a normal...
-
State the order of decisions in method development for gas chromatography.
-
The plaintiffs, Lee and Yoon, were the parents of H.L., a South Korean citizen who attended high school in Idaho as part of an exchange program. With permission from both plaintiffs and his host...
-
Amy Rockwell was a brilliant but penniless electrical engineer. She had designed a new type of cogeneration plant that she believed had great commercial potential. On January 15, she approached...
-
K Use appropriate formulas to find (a) the perimeter and (b) the area of the figure. 8.8 m 5 m 5.2 m 7 m (a) The perimeter is (Type an integer or a decimal.)
-
In Exercises 110, approximate each number using a calculator. Round your answer to three decimal places. e -0.75
-
In Exercises 914, complete the table. Round projected populations to one decimal place and values of k to four decimal places. Country Pakistan 2010 Population (millions) 184.4 Projected 2050...
-
The man attempts to pull down the tree using the cable and small pulley arrangement shown. If the tension in AB is 60 lb determine the tension in cable CAD and the angle that the cable makes at the...
-
O Macmillan Learning Solid ammonium chloride, NH Cl, is formed by the reaction of gaseous ammonia, NH3, and hydrogen chloride, HCl. NH3 (g) + HCl(g) NHCl(s) A 6.00 g sample of NH3 gas and a 6.00 g...
-
The Rydberg equation gives us the mathematical relationship between: A) an explanation for the continuous range of energy values associated with atomic spectra B) wavelength and frequency C) electron...
-
18. If 5.0 moles of calcium reacts with excess oxygen as shown, 2 Ca(s) + O2(g) 2CaO(s) AHxn= -1,270 kJ a. The reaction releases 6,350 kJ. b. The reaction absorbs 6,350 kJ. c. The reaction releases...
-
When aluminum is placed in concentrated hydrochloric acid, hydrogen gas is produced. 2 Al(s) + 6 HCl(aq) ->> 2 AICI, (aq) + 3 H(g) What volume of H(g) is produced when 7.20 g Al(s) reacts at STP?...
-
Draw a roadmap and write a plan to find the mass ( g ) of the products based on the given amount of reactants. 2 H 2 O 2 - > 2 H 2 O + O 2 given: H 2 O 2 = 4 9 . 4 mg
-
The predominant mode of decay for the charged pions is into a muon and a neutrino. The positive pion results in the neutrino and the negative pion in the antineutrino. (a) Write the decay equations...
-
Suppose that you could invest in the following projects but have only $30,000 to invest. How would you make your decision and which projects would you invest in? Project Cost $ 8,000 11,000 9,000...
-
A baseball is thrown downward with an initial velocity of 30 feet per second from a stadium seat that is 80 feet above the ground. Estimate to the nearest tenth of a second how long it takes for the...
-
If air resistance is ignored, the height h of a projectile above the ground after x seconds is given by h(x) = -1/2gx + v 0 x + h 0 where g is the acceleration due to gravity. This formula is also...
-
The number of unique monthly visitors in millions to Facebook can be approximated by V(x) = 16x 2 + 7x + 32, where x is the number of years after 2008. Estimate the year when Facebook averaged 55...
-
You are buying a ( $ 2 0 , 0 0 0 ) boat with a down - payment of ( $ 4 , 0 0 0 ) and you finance the remaining amount with a 3 year loan at ( 7 . 8 % ) compounded weekly. ( A ) What are your weekly...
-
Cost of Goods Sold, Profit margin, and Net Income for a Manufacturing Company The following information is available for Bandera Manufacturing Company for the month ending January 31: Cost of goods...
-
Suppose that a 5 year coupon bond of face value $ 100 and coupon rate 3% sells for $ 98 dollars, and that a 5 year coupon bond of face value $100 and coupon rate 7 % sells for $ 102. Both bonds pay...

Study smarter with the SolutionInn App


