In Exercises 58, the graph of a quadratic function is given. Write the functions equation, selecting from
Question:
In Exercises 5–8, the graph of a quadratic function is given. Write the function’s equation, selecting from the following options.
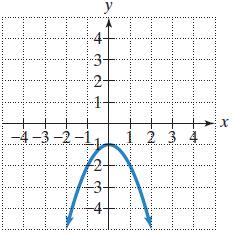
Transcribed Image Text:
f(x) = x² + 2x + 1 2 h(x) = x² - 1 g(x) = x² - 2x + 1 j(x) = -x² - 1
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (2 reviews)
The graph ...View the full answer

Answered By

Aun Ali
I am an Associate Member of Cost and Management Accountants of Pakistan with vast experience in the field of accounting and finance, including more than 17 years of teaching experience at university level. I have been teaching at both undergraduate and post graduate levels. My area of specialization is cost and management accounting but I have taught various subjects related to accounting and finance.
5.00+
13+ Reviews
32+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Mathematics questions
-
In each exercise, the graph of a quadratic function is given. Use the graph to find the solution set of each equation or inequality. (a) 3x + 10x8 = 0 (b) 3x+ 10x - 80 (c) 3x + 10x -8 <0 4 02111 FEB...
-
In each exercise, the graph of a quadratic function is given. Use the graph to find the solution set of each equation or inequality. (a) (b) (c) -3 2x - x + 15 = 0 2x-x+ 15 0 2x - x + 15 0 10 .0...
-
In each exercise, the graph of a quadratic function is given. Use the graph to find the solution set of each equation or inequality. (a) x + 3x + 10 = 0 (b) x + 3x + 10 0 (c) x + 3x + 10 0 F y 10...
-
b) A firm produces two types of sugar, A and B at a constant average cost of RM 2 and RM3 per kilogram, respectively. The quantities, q and qg (in kilogram) of A and B that can be sold each week are...
-
The following are six observations collected from treatment 1, ten observations collected from treatment 2, and eight observations collected from treatment 3. Test the hypothesis that the treatment...
-
(a) Would you expect d-glucaric acid to be optically active? (b) Write the open-chain structure for the aldaric acid (mannaric acid) that would be obtained by nitric acid oxidation of d-mannose. (c)...
-
A company wishes to hedge its exposure to a new fuel whose price changes have a 0.6 correlation with gasoline futures price changes. The company will lose $1 million for each 1 cent increase in the...
-
On June 1, 2014, Ellie Hopkins established an interior decorating business, First-Class Designs. During the month, Ellie completed the following transactions related to the business: June 1. Ellie...
-
I am facing a Database connection error (2). Could not connect to MySQL. error. Source codes are: Database Error Error establishing a database connection. Database connection error (2). Could not...
-
Fill in each blank so that the resulting statement is true. True or false: If the degree of the numerator of a rational function equals the degree of the denominator, then setting y equal to the...
-
In Exercises 18, use the Rational Zero Theorem to list all possible rational zeros for each given function. f(x) = x = x - 7x + 7x - 12x - 12 -
-
Fire frequency has a great impact on national parks. If the fire frequency is too severe, generally species that occur in later successional habitats will be excluded. Likewise, the absence of fire...
-
If your % Al is less than 99%, explain two possible sources of uncertainty that could account for your low value. If your % Al is greater than 100%, explain two possible sources of uncertainty that...
-
An excess of sodium hydroxide is treated with 26.5 L of dry hydrogen chloride gas measured at STP. What is the mass of sodium chloride formed? 1.55 kg 1.69 g 0.138 kg O69.1 g 13.3 g
-
In terms of the HSAB concept, which end of the SCN ion (S or N) would coordinate to Cr+ and Pt2+? Explain briefly
-
How many moles of Ca(OH)2 are needed to completely react with 440.6mL of 0.5M HNO3 solution
-
The Michealis Menten mechanism assumes that: The catalysed reaction is always zero order kinetics and the rate of the reaction does not depend on the concentration of the substrate ever. The...
-
Ellerson Company provided the following information for the last calendar year: Beginning inventory: Direct materials ...... $68,000 Work in process ...... 29,400 Finished goods ...... 43,200 Ending...
-
The first law of thermodynamics is sometimes whimsically stated as, You cant get something for nothing, and the second law as, You cant even break even. Explain how these statements could be...
-
Find the standard equation of a circle with center (-2, 3) and radius 7.
-
Find the point-slope form of the line passing through the given points. Use the first point as (x 1 , y 1 ). Then convert the equation to slope-intercept form and write a formula for a function f...
-
Let a 0. Solve |ax + b| = 0.
-
How does emotional intelligence contribute to effective conflict resolution and negotiation strategies, enabling individuals to manage interpersonal conflicts constructively and navigate complex...
-
How can organizations leverage emotional intelligence as a cornerstone of ethical leadership, promoting integrity, empathy, and social responsibility in decision-making processes and stakeholder...
-
Please show full step solutions, the answers are given. page 9 of 17 Question 5 We have 25.0 mL of a 0.400 M solution of HA(aq). We titrate this solution with a 0.400 M solution of NaOH(aq). The pH...

Study smarter with the SolutionInn App


