Apply the quotient rule for exponents, if possible, and write each result using only positive exponents. Assume
Question:
Apply the quotient rule for exponents, if possible, and write each result using only positive exponents. Assume that all variables represent nonzero real numbers.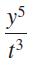
Transcribed Image Text:
13
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)
The quotient rule for exponents states that for any nonzero real numbers y and t and positive ...View the full answer

Answered By

ROXAN VEGA
I have extensive hands-on experience and proficiency in assisting students in various subjects. During my tutoring sessions, I prioritize creating a supportive and engaging learning environment. I strive to understand each student's unique learning style and tailor my teaching methods accordingly. I am patient, empathetic, and skilled at breaking down complex concepts into simpler, more manageable parts. I encourage active participation and discussion, promoting critical thinking and problem-solving skills.
One of my strengths as a tutor is my ability to adapt to different learning needs and styles. I can explain difficult concepts using real-life examples, visual aids, or interactive exercises, depending on what works best for the student. I also provide clear explanations, answer questions, and offer guidance in a clear and concise manner.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Intermediate Algebra
ISBN: 9780134895987
13th Edition
Authors: Margaret Lial, John Hornsby, Terry McGinnis
Question Posted:
Students also viewed these Mathematics questions
-
Apply the quotient rule for exponents, if possible, and write each result using only positive exponents. Assume that all variables represent nonzero real numbers. S 5-8 S
-
Apply the quotient rule for exponents, if possible, and write each result using only positive exponents. Assume that all variables represent nonzero real numbers. x y
-
Apply the quotient rule for exponents, if possible, and write each result using only positive exponents. Assume that all variables represent nonzero real numbers. -3
-
Write a Summary of Tartuffe the Norton anthology of western literature.
-
Which of the entrepreneurial characteristics mentioned in the chapter describe you? Which of these characteristics can you develop more fully in advance of running your own business?
-
Campbell Inc. produces and sells outdoor equipment. On July 1, 20Y1, Campbell issued $30,000,000 of 10-year, 10% bonds at a market (effective) interest rate of 9%, receiving cash of $31,951,110....
-
What types of remedies an aggrieved party may seek from the court?
-
You are the chief accountant of Deighton plc, which manufactures a wide range of building and plumbing fittings. It has recently taken over a smaller unquoted competitor, Linton Ltd. Deighton is...
-
1. Below is some data about the tshirt and jeans sales of a company. The company decided to raise their prices at the end of Q2. Item Tshirt Q1 Sales Jeans 150 @ $10.00 108 @ $25 Q2 Sales 152 @...
-
The tables give some selected ordered pairs for functions and g. Tables like these can be used to evaluate composite functions. For example, to evaluate (g ) (6), use the first table to find (6) =...
-
The tables give some selected ordered pairs for functions and g. Tables like these can be used to evaluate composite functions. For example, to evaluate (g ) (6), use the first table to find (6) =...
-
A food company is attempting to set the customer service level (in-stock probability in its warehouse) for a particular product line item. Annual sales for the item are 100,000 boxes, or 3,846 boxes...
-
Calculate the Debye length for a \(0.1-\mathrm{M}\) solution of a univalent \((1,1)\) electrolyte, i.e., one with single-charged cations and single-charged anions, in water. If the surface has a...
-
Extend the analysis of a system for transport in a charged membrane to the case of a mixture of two electrolyte salts with a common anion (a mixture of \(\mathrm{NaCl}\) and \(\mathrm{KCl}\), for...
-
Consider a charged membrane of thickness \(100 \mu \mathrm{m}\) with concentrations of \(1 \mathrm{M} \mathrm{HCl}\) on one side and \(0.1 \mathrm{M} \mathrm{HCl}\) on the other side. The membrane...
-
Find the flow rate for an electro-osmotic flow in a tube of diameter \(5 \mu \mathrm{m}\) and length \(10 \mathrm{~cm}\) filled with an aqueous solution. The applied potential is \(1 \mathrm{kV}\)....
-
Analyze the problem of electro-osmotic flow with an additional imposed pressure gradient. Show that the volumetric flow rate can be expressed as \[Q=L_{11} \Delta \phi+L_{12} \Delta P\] where...
-
Explain how a dictionary and thesaurus can help you be a more effective communicator.
-
For the following exercises, rewrite the sum as a product of two functions or the product as a sum of two functions. Give your answer in terms of sines and cosines. Then evaluate the final answer...
-
A store sells refurbished iPhones that cost 12% less than the original price. If the new price of a refurbished iPhone is $572, what was the original price? How much is saved by purchasing the...
-
A car dealer, at a year-end clearance, reduces the list price of last years models by 15%. If a certain four-door model has a discounted price of $18,000, what was its list price? How much can be...
-
How many right triangles have a hypotenuse that measures 2x + 3 meters and legs that measure 2x - 5 meters and x + 7 meters? What are their dimensions?
-
Darlene Saunders has carried on a business as a sole proprietor for over 10 years. The business has been very successful and has now reached a point where it is producing more income than she...
-
b) Later this semester, we will derive the Maxwell-Boltzmann speed distribution P(v) for molecules with 2 m mass m at temperature T, P(v) = 4 exp 2kBT my 2kRT B where kB is Boltzmann's constant....
-
Juice level (cm) 1. Finley filled an empty drink dispenser with juice. The juice level, in centimeters, is proportional to time elapsed, in minutes, since she began filling the drink dispenser. The...

Study smarter with the SolutionInn App


