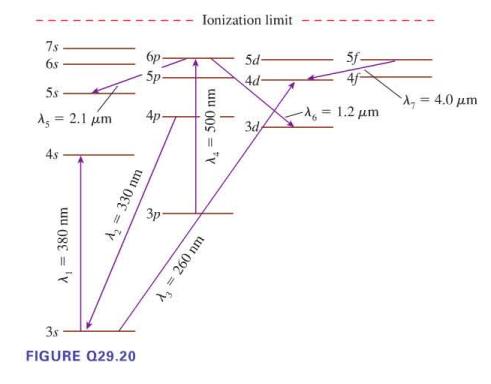
Seven possible transitions are identified on the energy level diagram in Figure Q29.20. For each, is this
Question:
Seven possible transitions are identified on the energy level diagram in Figure Q29.20. For each, is this an allowed transition? If allowed, is it an emission or an absorption transition, and is the photon infrared, visible, or ultraviolet? If not allowed, why not?
Transcribed Image Text:
7s 68 5s A = 2.1 μm 4s A₁ = 380 nm A₂ = 330 nm 3.s FIGURE Q29.20 бр 5p- 3p Ionization limit A₂ = 500 nm A₂ = 260 nm 5d- 4d- 3d/ 5f- -λ = 1.2 μm = 4.0 μm
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
Here is an analysis of the seven possible transitions identified on the energy level diagram in Figure Q2920 Transition Allowed Type of Transition Typ...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

College Physics A Strategic Approach
ISBN: 9780321595492
2nd Edition
Authors: Randall D. Knight, Brian Jones, Stuart Field
Question Posted:
Students also viewed these Physics questions
-
Figure 28.19 shows part of the energy level diagram for the electrons in an imaginary atom. The arrows represent three transitions between the energy levels. For each of these transitions: a....
-
Question 1 [34 marks] Read the scenarios below for the year ending 28 February 2023 and answer the following question. A. The following is the income statement of Axe Ltd for the financial year...
-
In a set of experiments on a hypothetical one-electron atom, you measure the wavelengths of the photons emitted from transitions ending in the ground state (n = I), as shown in the energy level...
-
Were Napoleon's territorial ambitions significantly different from pre-1799 conquests? If so, where?What were Napoleon's most significant domestic accomplishments in France? Consider the interesting...
-
Zippy Computers announced strong fourth quarter results. Sales and earnings were both above analysts expectations. You notice in the newspaper that Zippys stock price went up sharply on the day of...
-
McCurdy Oil acquired an existing oil well and all related equipment used in the production of oil. McCurdy paid \($2\),500,000, of which 20% was attributable to pumps, pipelines, and tanks. The oil...
-
The Foreign Corrupt Practices act can levy fines of more than five hundred million against companies our size that do not eliminate corruption in their operations abroad. Can you understand why the...
-
The following information relates to depreciable assets of Strata Technologies. (a) Machine A was purchased for $80,000 on January 1, 2006. The entire cost was erroneously expensed in the year of...
-
What are some of the most effective ways that companies can detect and prevent inventory and noncash assets fraud? How can companies establish robust internal controls over their inventory and asset...
-
List the quantum numbers of (a) All possible 3p states (b) All possible 3d states.
-
Show, by actual calculation, that the Bohr radius is 0.0529 nm and that the ground-state energy of hydrogen is -13.60 eV.
-
An airline has 200 seats in the coach portion of the cabin of an Airbus A340. It is attempting to determine how many seats it should sell to business travelers and how many to vacation travelers on a...
-
Monthly temperatures: 63F, 68F, 73F, 78F, and 83F Determine the level of measurement of each variable.
-
A quality-control manager randomly selects 70 bottles of ketchup that were filled on July 17 to assess the calibration of the filling machine. A research objective is presented. For each, identify...
-
Suppose you are conducting a survey regarding violence in hockey in a youth hockey league. You obtain a cluster sample of 10 hockey teams within a youth hockey league and sample all hockey players in...
-
Let \(Y\) be distributed according to the gamma(12 4) distribution. (a) Find \(\mathrm{E}[Y]\). (b) Find \(\operatorname{Var}[Y]\). (c) Find \(P(Y\)
-
The average score for a class of 28 students taking a calculus midterm exam was 72%. Determine whether the underlined value is a parameter or a statistic.
-
On May 1, 2012, Lakeville Company bought a patent at a cost of $8,400. It is estimated that the patent will give Lakeville a competitive advantage for 10 years. Record in general journal form...
-
Use critical values to test the null hypothesis H0: 1 2 = 20 versus the alternative hypothesis H0: 1 2 20 by setting a equal to .10, .05, .01, and .001. How much evidence is there that the...
-
A rocket in deep space has an exhaust-gas speed of 2000 m/s. When the rocket is fully loaded, the mass of the fuel is five times the mass of the empty rocket. What is the rockets speed when half the...
-
A tennis player swings her 1000 g racket with a speed of 10 m/s. She hits a 60 g tennis ball that was approaching her at a speed of 20 m/s. The ball rebounds at 40 m/s. a. How fast is her racket...
-
A 60 g tennis ball with an initial speed of 32 m/s hits a wall and rebounds with the same speed. FIGURE P11.39 shows the force of the wall on the ball during the collision. What is the value of F max...
-
Data Exploration - connect - CH 5 Understanding Risk For detailed instructions on using Federal Reserve Economic Data (FRED) online to answer each of the following problems, visit...
-
M3 | Investments Basics I: Financial Services, Stocks, and Mutual Funds Identify three factors that are more important to you for selecting a financial institution. Using the websites of three...
-
My Annual Comprehensive Financial Report is on Delaware state and pls include references. 1. Explore the concepts of the course material relating to ACFRs. You must also find and review / read...

Study smarter with the SolutionInn App


