Two vectors appear as in Figure Q3.18. Which combination points directly to the left? TA FIGURE Q3.18
Question:
Two vectors appear as in Figure Q3.18. Which combination points directly to the left?

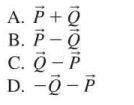
Transcribed Image Text:
TA FIGURE Q3.18 ē
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
Answer The correct answer is option C Explanation The combination of vectors is shown ...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

College Physics A Strategic Approach
ISBN: 9780321595492
2nd Edition
Authors: Randall D. Knight, Brian Jones, Stuart Field
Question Posted:
Students also viewed these Physics questions
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
The Crazy Eddie fraud may appear smaller and gentler than the massive billion-dollar frauds exposed in recent times, such as Bernie Madoffs Ponzi scheme, frauds in the subprime mortgage market, the...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Consider the following excerpts from a Wall Street Journal article on Burger King (Beatty, 1996): Burger King intends to bring smiles to the faces of millions of parents and children this holiday...
-
In the Arnold's Muffler example for the exponential distribution in this chapter, the average rate of service was given as 3 per hour, and the times were expressed in hours. Convert the average...
-
The accounting staff of CCB Enterprises has completed the financial statements for the 2014 calendar year. The statement of income for the current year and the comparative statements of financial...
-
A structural member in a nuclear reactor is made of a zirconium alloy. If an axial load of 4 kip is to be supported by the member, determine its required cross-sectional area. Use a factor of safety...
-
Identify in what ledger (general or subsidiary) each of the following accounts is shown. 1. Rent Expense 2. Accounts ReceivableChar 3. Notes Payable 4. Accounts PayableThebeau
-
Punishments typically identified as intermediate sanctions include intensive supervision probation (ISP), drug courts, fines, community service, day reporting centers, remote-location monitoring,...
-
A car traveling at 30 m/s runs out of gas while traveling up a 5.0 slope. How far will it coast before starting to roll back down?
-
You begin sliding down a 15 ski slope. Ignoring friction and air resistance, how fast will you be moving after 10 s?
-
If is sufficient for , show that any one-to-one function of is also sufficient for .
-
Where along the long-run average total cost curve will an efficient firm try to produce in the long run?
-
Assign a measure of utility to your studying for various courses. Do your study habits follow the principle of rational choice?
-
What are the monopolistic and the competitive elements of monopolistic competition?
-
Often, people buy a good to impress others and not because they want it. a. What implications would such actions have for the application of economic analysis? b. How many goods are bought because...
-
What are the ways in which a firm can differentiate its product from that of its competitors? What is the overriding objective of product differentiation?
-
Enter the account names and dollar amounts from the comparative balance sheets in Exhibit13.1 into a worksheet in a spreadsheet file. Create a second copy of the worksheet in the same spread-sheet...
-
A routine activity such as pumping gasoline can be related to many of the concepts studied in this text. Suppose that premium unleaded costs $3.75 per gal. Work Exercises in order. Use the...
-
The batteries shown in the circuit in Fig. E26.24 have negligibly small internal resistances. Find the current through (a) The 30.0- resistor; (b) The 20.0- resistor; (c) The 10.0-V Figure E26.24...
-
You are working late in your electronics shop and find that you need various resistors for a project. But alas, all you have is a big box of 10.0- resistors. Show how you can make each of the...
-
A resistor with R 1 = 25.0 is connected to a battery that has negligible internal resistance and electrical energy is dissipated R 1 by at a rate of 36.0 W. If a second resistor with R 2 = 15.0 is...
-
Plutonium-239 has a half-life of approx. 24, 120 years. Assuming we started with 30 grams, how many grams of plutonium would there still be after 39,056 years?
-
In 2024, the Westgate Construction Company entered into a contract to construct a road for Santa Clara County for $10,000,000. The road was completed in 2026. Information related to the contract is...
-
Nitrogen monoxide can be synthesized by combining nitrogen and oxygen under certain conditions: N2(g) + O2(g) = 2NO(g) At 1500K, the reaction has an equilibrium constant of 1.10x10-5. If air is...

Study smarter with the SolutionInn App


