A piece of gold has a mass of 20 g. If this gold were formed into a
Question:
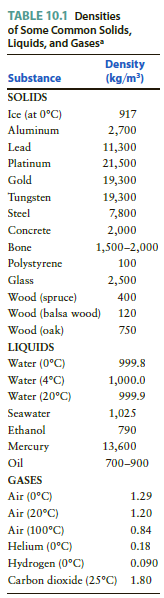
Transcribed Image Text:
TABLE 10.1 Densities of Some Common Solids, Liquids, and Gases Density (kg/m) Substance SOLIDS Ice (at 0°C) Aluminum 917 2,700 Lead 11,300 Platinum 21,500 Gold 19,300 Tungsten 19,300 Steel 7,800 Concrete 2,000 Bone 1,500-2,000 Polystyrene 100 Glass 2,500 Wood (spruce) Wood (balsa wood) 400 120 Wood (oak) 750 LIQUIDS Water (0°C) 999.8 Water (4°C) 1,000.0 Water (20°C) 999.9 Seawater 1,025 Ethanol 790 13,600 Mercury Oil 700-900 GASES Air (0°C) 1.29 Air (20°C) 1.20 Air (100°C) 0.84 Helium (0°C) 0.18 Hydrogen (0°C) 0.090 Carbon dioxide (25°C) 1.80
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (15 reviews)
Apply the concepts of density mass and volume No sketch needed The density of gold as given in Table ...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

College Physics Reasoning and Relationships
ISBN: 978-0840058195
2nd edition
Authors: Nicholas Giordano
Question Posted:
Related Video
In this video, A mixture of methanol and air in a large polycarbonate bottle is ignited. The resulting rapid combustion reaction, often accompanied by a dramatic ‘whoosh’ sound and flames, demonstrates the large amount of chemical energy released in the combustion of alcohol
Students also viewed these Sciences questions
-
You have a piece of gold jewelry weighing 9.35 g. Its volume is 0.654 cm3. Assume that the metal is an alloy (mixture) of gold and silver, which have densities of 19.3 g/cm3 and 10.5 g/cm3,...
-
Gold is alloyed (mixed) with other metals to increase its hardness in making jewelry. (a) Consider a piece of gold jewelry that weighs 9.85 g and has a volume of 0.675 cm3. The jewelry contains only...
-
A piece of gold of mass 0.250 kg and at a temperature of 75.0C is placed into a 1.500-kg copper pot containing 0.500 L of water. The pot and water are at 22.0C before the gold is added. What is the...
-
Which of the following is NOT a factor to be considered in determining a limited-life intangible assets useful life? The expected useful life of any related asset All of the other answers are correct...
-
In the previous problem, suppose the required return on the project is 14 percent. What is the project's NPV? In the previous problem Cochrane, Inc., is considering a new three-year expansion project...
-
What risks would be found if the Maser Plan incorporated and Expansion out of state?
-
Meddevco (name changed) is a large multinational corporation that operates in the medical devices sector. The firm employs around 33,000 people in five divisions and has operations in 120 countries....
-
Discuss why variable pay-for-performance plans have become popular and what elements are needed to make them successful.
-
A 50-ft diameter anaerobic digester with 10-ft side-wall depth treats 30,000 gpd of mixed primary and secondary sludge. The mixed sludge has a 4% total solids concentration and is 75% volatile...
-
Directions: For each of the definitions, write the letter of the appropriate term in the space provided. a. Help system b. Option buttons c. Check boxes d. Command buttons e. Window f. Drop-down menu...
-
In a neutron star, all the mass has collapsed into a relatively small volume. (a) If a particular neutron star has a radius of 1000 m and a mass of 2.0 10 28 kg, what is its density? (b) Compare...
-
Figure Q10.4 is a photograph of a graduated cylinder filled with four fluids. Starting with mercury on the bottom and going up, we have salt water, water, and vegetable oil. In addition, a solid...
-
A company reports the following: Net income ..........$1,000,000 Preferred dividends ........ 50,000 Average stockholders equity ... 6,250,000 Average common stockholders equity. 3,800,000 Determine...
-
Discuss and give example how deontology and consequentialism relates to consent in nursing profession.
-
You will identify a problem or issue in your present or past workplace, or in your community. The assignment objective as provided in Unit 4, is to propose a solution to the problem or issue, and to...
-
What are factoids and can you give an example of how they are used today?
-
What are the benefits of Artificial Intelligence in healthcare?
-
Under line sensory words. The Big Mac is a juicy, delicious burger that will leave you feeling satisfied. The bun is soft and fluffy, and the beef is cooked to perfection. The secret sauce is the...
-
Based on the StokesEinstein equation, how does the diffusion coefficient for a particle depend on the size of the particle?
-
Match the following. Answers may be used more than once: Measurement Method A. Amortized cost B. Equity method C. Acquisition method and consolidation D. Fair value method Reporting Method 1. Less...
-
Why is atomic level resolution obtained on pentacene in the AFM mode as shown in Figure 16.18, but not in the STM mode? Figure 16.18 1.3 A OA +1Hz -2Hz D 20A 5A -7Hz. -5Hz.
-
Why were quantum dots emitting in the near infrared region used for the surgery experiment shown in Figure 16.25? Figure 16.25 Color video 5 min post-injection NIR fluorescence 5 min post-injection...
-
In this problem, you will calculate the transmission probability through the barrier illustrated in Figure 16.10. We first go through the mathematics leading to the solution. You will then carry out...
-
Paid Phoenix Insurance $50 for February renters insurance using company VISA credit card. Record as Renter Insurance Expense. What are the Accounts and Amounts Debited? What are the Accounts and...
-
Discuss employers' responsibilities for reporting workplace incidents that result in serious injury or illness. Include in your discussion the time frames for this reporting, whether or not you...
-
Which 60" aspect ratio resource is a larger screen area 4.3 or 16.9?

Study smarter with the SolutionInn App


