What is the quantum number of this hydrogen atom?
Question:
What is the quantum number of this hydrogen atom?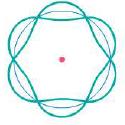
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
n 3 Each antinode is ...View the full answer

Answered By

Jehal Shah
I believe everyone should try to be strong at logic and have good reading habit. Because If you possess these two skills, no matter what difficult situation is, you will definitely find a perfect solution out of it. While logical ability gives you to understand complex problems and concepts quite easily, reading habit gives you an open mind and holistic approach to see much bigger picture.
So guys, I always try to explain any concept keeping these two points in my mind. So that you will never forget any more importantly get bored.
Last but not the least, I am finance enthusiast. Big fan of Warren buffet for long term focus investing approach. On the same side derivatives is the segment I possess expertise.
If you have any finacne related doubt, do reach me out.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

College Physics A Strategic Approach
ISBN: 9780321907240
3rd Edition
Authors: Randall D. Knight, Brian Jones, Stuart Field
Question Posted:
Students also viewed these Sciences questions
-
A hydrogen atom (Z = 1) is in the third excited state, and a photon is either emitted or absorbed. Concepts: (i) What is the quantum number of the third excited state? (ii) When an atom emits a...
-
A hydrogen atom emits a photon that has momentum with a magnitude of 5.452 x 10-27 kg ? m/s. This photon is emitted because the electron in the atom falls from a higher energy level into the n = 1...
-
Write a program in your favorite programming language (Python, C, C++, etc.) to implement/simulate a finite automaton that (only) recognizes/accepts the language of: Problem A Binary numbers...
-
The programming language is Java and all of the Classes I was given are in bold. ALIEN CLASS import imagePackage.RasterImage; import java.awt.BasicStroke; import java.awt.Color; import...
-
Suppose the following financial data were reported by 3M Company for 2018 and 2019 (dollars in millions). Instructions (a) Calculate the current ratio and working capital for 3M for 2018 and 2019....
-
Describe the legal features of fee simple absolute.
-
For each of the situations listed, identify the primary standard from the IMA Statement of Ethical Professional Practice that is violated (competence, confidentiality, integrity, or credibility)....
-
Direct manufacturing labor and direct materials variances, missing data. (CMA heavily adapted). More Bay Surfboards manufactures fiberglass surfboards. The standard cost of direct materials and...
-
Suppose that the supply curve of serving in the military is S(P) = P, where Q = S(P) is several hours employed in military service a day. There are only mercenaries in the army (there is no draft). P...
-
Write the symbol for an atom or ion with: a. four electrons, four protons, and five neutrons. b. six electrons, seven protons, and eight neutrons.
-
How many electrons, protons, and neutrons are contained in the following atoms or ions: (a) \({ }^{9} \mathrm{Be}^{+}\), (b) \({ }^{12} \mathrm{C}\), and (c) \({ }^{15} \mathrm{~N}^{+++}\)?
-
An insulin receptor substrate (IRS) contains a so-called plekstrin homology (PH) domain that binds to the inositol head group of membrane lipids, a phosphotyrosine-binding (PTB) domain that differs...
-
A company has $90,000 in outstanding accounts receivable and it uses the allowance method to account for uncollectible accounts. Experience suggests that 4% of outstanding receivables are...
-
What type of force, either the applied force, frictional force, or sum of forces that really effects the acceleration of an object. Explain.
-
A project requires an initial investment of $50,000. The project will generate net cash flows of $15,000 at the end of the first year, $40,000 at the end of the second year, and $10,000 at the end of...
-
Nebraska Inc. issues 4,750 shares of common stock for $152,000. The stock has a stated value of $14 per share. The entry to journalize the stock issuance would include a credit to Common Stock for
-
If an employee option to buy 4,000 shares of the employer's stock has a strike price of $15.50 and on May 29, 2021 the stock's market price is $29.90, what is the intrinsic value of this option?...
-
Let us model the electromagnetic wave in a microwave oven as a plane traveling wave moving to the left, with an intensity of 25.0 kW/m2. An oven contains two cubical containers of small mass, each...
-
This problem continues the Draper Consulting, Inc., situation from Problem 12-45 of Chapter 12. In October, Draper has the following transactions related to its common shares: Oct 1 Draper...
-
Mortgage Division: Often a mortgage payment stream is divided into a principal payment stream and an interest payment stream and the two streams are sold sepa- rately. Consider a standard mortgage of...
-
Cruz Company uses LIFO for inventory costing and reports the following financial data. It also recomputed inventory and cost of goods sold using FIFO for comparison purposes. Year 2 Year 1 LIFO...
-
Eastport Inc. was organized on June 5, Year 1. It was authorized to issue 300,000 shares of $10 par common stock and 50,000 shares of 5 percent cumulative class A preferred stock. The class A stock...

Study smarter with the SolutionInn App


