Estimate the energy released in the fission reaction of Eq. (29-48) from the values of the binding
Question:
Estimate the energy released in the fission reaction of Eq. (29-48) from the values of the binding energy per nucleon in Fig. 29.2.
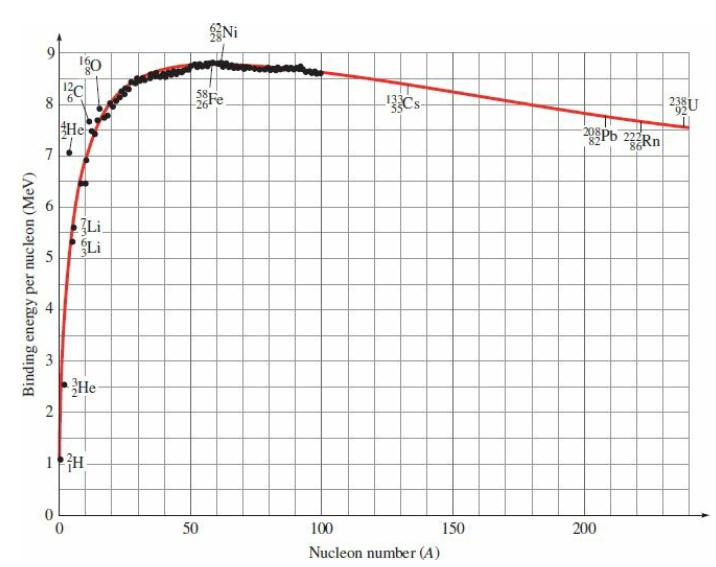
![]()
Transcribed Image Text:
8 7 5 Binding energy per nucleon (MeV) 3 + go ¹2C He Li He 2 1 H Ni 58 Fe 50 133 100 Nucleon number (A) 150 208Pb 22Rn 86 200 20
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (10 reviews)
The energy released in the fission reaction of Eq 2948 can be estimated from the values of the bindi...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

College Physics With An Integrated Approach To Forces And Kinematics
ISBN: 978-1260547719
5th Edition
Authors: Alan Giambattista
Question Posted:
Students also viewed these Sciences questions
-
Estimate the energy released in the fission reaction of Eq. (29-31). Look up the binding energy per nucleon of the nuclides in Fig. 29.2.
-
What is the energy released in the fission reaction of Eq. 31-4? (The masses of 141/56Ba and 92/36Kr are 140.914411u and 91.926156u. respectively).
-
Calculate the energy released in the fission reaction of Eq. (29-30). The atomic masses of 14156Ba and 9236Kr are 140.914 u and 91.926 u, respectively.
-
Mr. Yap bought a bond having a face value of Php 1,000.00 for Php 970.00. The bond rate was 14% nominal and dividends were made to him semiannually for a total of 7 years. At the end of the seventh...
-
Are the events Wednesday and in the fifth week independent? Explain. Sun Mon Tues Wed Thurs Fri Sat 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30
-
Seneca Co. began year 2015 with 6,500 units of product in its January 1 inventory costing $35 each. It made successive purchases of its product in year 2015 as follows. The company uses a periodic...
-
A vertical plate \(3 \mathrm{~m}\) long is at a temperature of \(400 \mathrm{~K}\) and exposed to air at \(300 \mathrm{~K}\). Calculate the thickness of the boundary layer and the value of the local...
-
When one company buys another company, it is not unusual that some workers are terminated. The severance benefits offered to the laid-off workers are often the subject of dispute. Suppose that the...
-
A company plans to make four annual deposits of $ 3 , 7 5 0 each to a special building fund. The fund s assets will be invested in mortgage instruments expected to pay interest at 1 2 % on the fund s...
-
On June 1, 2019, Kris Storey established an interior decorating business, Eco-Centric Designs. During the month, Kris completed the following transactions related to the business: June 1. Kris...
-
Calculate the energy released in the fission reaction of Eq. (29-47). on + 235U 235U* 230U* 14Ba + Kr + 3n (29-47)
-
The greatest concentration of iodine in the body is in the thyroid gland, so radioactive iodine-131 is often used as a tracer to help diagnose thyroid problems. Suppose the activity of 131I in a...
-
Contrast the following terms: a. Break-even analysis; present value; net present value; return on investment b. Economic feasibility; legal and contractual feasibility; operational feasibility;...
-
2. A pendulum is raised 0.70m above the floor, and then is released and swings freely. Neglecting friction forces, what would be its velocity as it swings at a point 0.25m above the floor?
-
A negatively charged Teflon rod is brought near a neutral conductor. Does the rod exert a net force on the conductor? If so, in what direction is the force on the conductor? Select One of the...
-
You land a great job at GE. Your starting pay was $87,000. With raises and a promotion, six years later, your pay is $117,438. Use this information to compute the values represented by A and B in the...
-
6) The Crescent Street Jam (a local jazz quintet, with your Professor on Piano) has market power. Consumers in Potsdam demand tickets to each live performance according to P=18-0.2*Q (MR=18-0.4Q)....
-
3. A manufacturing company has the production capacity of 10,000 units per annum. The expenses for production of 5,000 (50%) units for a period are given as follows: Prepare levels P Particulars...
-
How is positional feedback obtained in robots?
-
What is a lobbyist in US? How did this term emerge?
-
For most ketones, hydrate formation is unfavorable, because the equilibrium favors the ketone rather than the hydrate. However, the equilibrium for hydration of hexafluoroacetone favors formation of...
-
Draw a plausible mechanism for each of the following transformations: (a) (b) (c) (d) Meo, OMe [H,SO4] excess MeOH -H20
-
Draw a plausible mechanism for each of the following reactions: (a) (b) [H,SO4] -H20 [H,SO4] -H20
-
What is the primary reason that auditors' assess internal control?
-
Alan Jones, a U.S. citizen, incorporates D Corp., a non-treaty Country X company, to do active business in Country X. There is no tax treaty between the U.S. and Country X. D Corp. has Country X...
-
In 2023, USCO, which is subject to a 21% U.S. corporate tax rate, has $1 Million of worldwide active income which is all from foreign sources in Country X. Assume USCO's foreign-source active income...

Study smarter with the SolutionInn App


