One possible fission reaction for 235 U is 235 U + n 141 Cs + 93
Question:
One possible fission reaction for 235U is 235U + n → 141Cs + 93Rb + ?n, where “?n” represents one or more neutrons.
(a) How many neutrons?
(b) From the graph in Fig. 29.2, you can read the
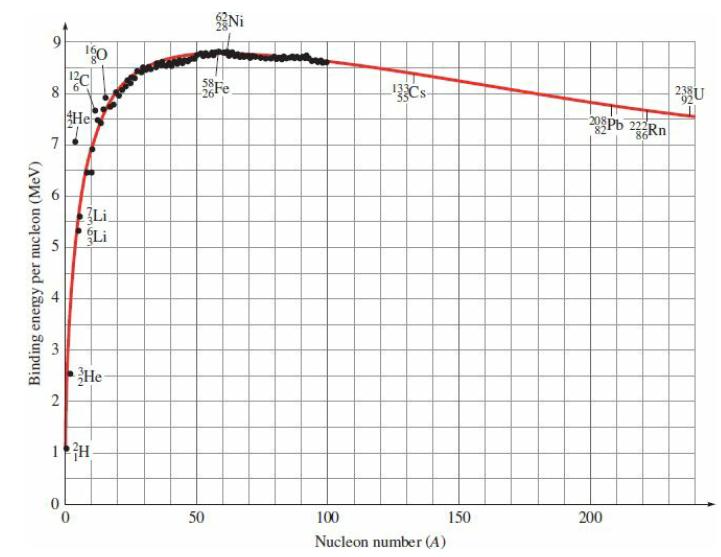
Transcribed Image Text:
∞ 7 5 Binding energy per nucleon (MeV) 3 4 26 Igo 12C He tut 2 1 H Li Li He- 50 Ni Fe 100 Nucleon number (A) 150 208Pb 222Rn 200 23 U
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (17 reviews)
a The number of neutrons produced in this fission reaction is not specified in ...View the full answer

Answered By

Akshay Shete
I have extensive experience as a tutor, both online and in-person. I have worked with students of all ages and abilities, and am skilled at adapting my teaching style to meet the needs of each individual student. I have a strong background in a variety of subjects, including math, science, and English, and am able to break down complex concepts in a way that is easy for students to understand. In addition to my subject matter expertise, I am also a patient and supportive teacher, and am committed to helping my students succeed. Whether I am working with a struggling student who needs extra help to catch up, or an advanced student looking to get ahead, I am able to provide the guidance and support they need to reach their goals. Overall, my hands-on experience as a tutor has prepared me to be a confident and effective teacher, and I am excited to use my skills to help students succeed.
0.00
0 Reviews
10+ Question Solved
Related Book For 

College Physics With An Integrated Approach To Forces And Kinematics
ISBN: 978-1260547719
5th Edition
Authors: Alan Giambattista
Question Posted:
Students also viewed these Sciences questions
-
One possible fission reaction for 235U is 235U + n 141Cs + 93Rb + ?n, where "?n" represents one or more neutrons. (a) How many neutrons? (b) From the graph in Fig. 29.2, you can read the approximate...
-
From the graph in the margin relating the holding- period returns for Aram Inc. to the S& P 500 Index, estimate the firms beta.
-
Find the adjacency matrix of the graph in Fig 476.
-
A total of 34,695 adults were asked whether they agreed with the following statement: Evolution is the best explanation for the origins of human life on earth. Table 19 compares the adults responses...
-
The world record of mens pole vault is 6.16 m while the female record is 5.06 m. What is the difference in these heights in feet? THINKING IT THROUGH. After using the correct conversion factor, the...
-
Fab and Fast Ltd buys and sells motor vehicle accessories. The firms estimated sales and expenses for the first 4 months of 2025 are shown below. Actual sales for December 2024 were $900000 and...
-
Yun Tung Chow tried to unclog a floor drain in the kitchen of the restaurant where he worked. He used a drain cleaner called Lewis Red Devil Lye that contained crystalline sodium hydroxide. The...
-
We have a non-rotating space station in the shape of a long thin uniform rod of mass 4.45 x 10^6 kg and length 1387 meters. Small probes of mass 9598 kg are periodically launched in pairs from two...
-
Consider an LSI medical imaging system with PSF given by h(x, y) = (1/ 2) e (x2+y2 )/2) (a) Calculate the MTF associated with this system. (b) Plot the MTF as a function of frequency. (c) If a...
-
What is the total energy released by the proton-proton cycle [Eq. (29- 54)]? 4p + 2e He + 2v
-
Calculate the energy released in the fission reaction of Eq. (29-47). on + 235U 235U* 230U* 14Ba + Kr + 3n (29-47)
-
The following is a December 31, 2013, post-closing trial balance for the Jackson Corporation. Required: Prepare a classified balance sheet for Jackson Corporation at December 31,2013. Account Title...
-
2.4 Draw the truth table of a 2-input XOR gate. (2) QUESTION 3. [30] 3.1 (a) Convert following SOP (Sum of Products) Boolean expression to a truth table: (5) X(ABC) ABC+ A B+ABC (b) Write the Boolean...
-
19. The riskless rate is 4.7%. There are five risky assets. Their expected returns are 11.3%, 7.8%, 9.2%, 13.6%, and 10.5%. Their standard deviations are 16.6%, 24.3%, 19.7%, 22.9%, and 18.3%. All of...
-
Without considering the following capital gains and losses, Carley, who is single, has taxable income of $325,000 and a marginal tax rate of 35%. During the year, she sold stock held for nine months...
-
The objective is to utilize ratio analysis to make financial decisions. ? We learned that ratios are used extensively in financial management to help determine the overall financial health of an...
-
ELLA'S ELECTRONICS ACCOUNTING PROJECT PART 2Adjusting InformationAt the end of February, the following information is determined:a. One month of the business insurance coverage has expired.b. At the...
-
If det A = 2, det B = -1, and det C = 3, find: (a) (b) det X B 0 (d) det 0 B 0
-
A certain Christmas tree ornament is a silver sphere having a diameter of 8.50 cm. Determine an object location for which the size of the reflected image is three-fourths the size of the object. Use...
-
Draw the structure of each of the following compounds: (a) (S)-3,3-dibromo-4-ethylcyclohexanone (b) 2,4-dimethyl-3-pentanone (c) (R)-3-bromobutanal
-
Provide a systematic (IUPAC) name for the compound below. Be careful: This compound has two chirality centers (can you find them?).
-
The compound above is an artificial flavor added to microwave popcorn and movietheater popcorn to simulate the butter flavor. Interestingly, this very same compound is also known to contribute to...
-
Suppose a male member currently aged 35, entered service at his age of 25, has a current salary of $75, 000 and total past salary of $650, 000. Assume that salaries increase at the beginning of each...
-
The Acme Rocket Car Company has a $9000 note due in 5 years. How much should be deposited at the end of each quarter in a sinking fund to pay off the note if the interest rate is 6.3%.
-
If you put $100 per month in account earning 3% annual interest, compounded monthly for 10 years, how much can you withdraw from that account each month for the next year?

Study smarter with the SolutionInn App


