Rank the six gases of Problem 43 in order of the total translational kinetic energy, greatest to
Question:
Rank the six gases of Problem 43 in order of the total translational kinetic energy, greatest to least.
Data From Problem 43
Six cylinders contain ideal gases (not necessarily the same gas) with the properties given (P = pressure, V = volume, N = number of molecules). Rank them in order of temperature, highest to lowest.
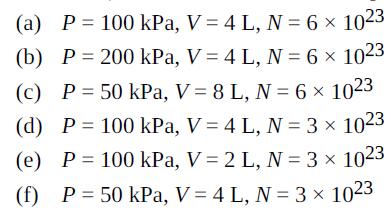
Transcribed Image Text:
(a) P = 100 kPa, V = 4 L, N = 6 x 1023 (b) P = 200 kPa, V = 4 L, N = 6 x 1023 (c) P= 50 kPa, V = 8 L, N = 6 x 1023 (d) P = 100 kPa, V = 4 L, N = 3 x 1023 (e) P = 100 kPa, V = 2 L, N = 3 x 1023 (f) P = 50 kPa, V = 4 L, N = 3 x 1023
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (6 reviews)
Strategy Expression for ideal gas equation Here is the pressure of the gas is the volume of the gas is the number of molecules is the temperature of t...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

College Physics With An Integrated Approach To Forces And Kinematics
ISBN: 978-1260547719
5th Edition
Authors: Alan Giambattista
Question Posted:
Students also viewed these Sciences questions
-
Rank the molecules below from lowest to highest according to their ability to diffuse across a lipid bilayer. Explain your rationale. H CCH CH2 NH H H3C NH
-
Rank the following six transactions from lowest to highest transaction costs. Explain your ranking by reference to the costs of search, bargaining, and enforcement. a. Getting married. b. Buying an...
-
Six cylinders contain ideal gases (not necessarily the same gas) with the properties given (P = pressure, V = volume, N = number of molecules). Rank them in order of temperature, highest to lowest....
-
1. Prepare program using threads in java that can print 10 times the numbers 1,2,3,4,5 in a series. 2. Prepare program using threads and a semaphore in java that can print the numbers 1,2,3,4,5 in a...
-
What total amount must be paid on July 4 to settle invoices dated June 20 for $485, June 24 for $367, and June 30 for $722, all with terms 11/2 /10, n/30?
-
A flexible exchange rate combined with a willingness to change the domestic interest rate can increase the effectiveness of monetary policy in an open economy. Consider an economy that suffers a fall...
-
Determine the stiffness matrix for a \([+45 /-45]\) antisymmetric laminate consisting of 0.25-mm-thick unidirectional AS/3501 carbon/epoxy plies.
-
Stylz Company, a recent start-up fashion retailer based in the United States, is deciding between opening its first sales presence in either Italy's Tuscany Region or Spain's Matarrana Region....
-
Evaluate the challenges and strategies associated with implementing quality control in complex, global supply chains, considering the impact of multi-tier suppliers, geographical dispersion, and...
-
Jay Rexford, president of Photo Artistry Company, was just concluding a budget meeting with his senior staff. It was November of 20x4, and the group was discussing preparation of the firms master...
-
What is the total translational kinetic energy of the gas molecules of air at atmospheric pressure that occupies a volume of 1.00 L?
-
Consider the expansion of an ideal gas at constant pressure. The initial temperature is T0 and the initial volume is V 0 . (a) Show that V/V 0 = T, where = 1/T 0 . (b) Compare the coefficient of...
-
Monkey see, Monkey do. Is this what is going on with drug prices. Read this link (https://www.bloomberg.com/news/articles/2015-10-02/pfizer-raised-prices-on-133-drugs-this-year-and-it-s-not-alone)...
-
FastJet Airlines Corp. is considering the cash purchase of four new airliners for $20,000,000 each. These airplanes will save the company a total of $300,000 per year in operational expenses compared...
-
While Cassidy is constrained in meeting her retirement objective due to limited cashflow, the Mensa's retirement goal is well within their means. Given their different financial situations, why might...
-
According to the plan presented by the company "El guila" to the Head of the "Los ingenios" Workshop, it is observed in the box and list of materials of the plan that the manufacture of 20 staggered...
-
Review the paralegalassociations (NFPA, NALS, NALA via the internet). Answer the following questions: What are the requirements to join (how can you become a member)? Do you possess any of the...
-
Are share repurchases common? What do these repurchases signal to the public?
-
Find the center of mass of that part of the cardioid of Problem 26 that is outside the circle r = a. Consider the lamina S of constant density k bounded by the cardioid r = a(1 + sin (), as shown in...
-
In the operation of an automated production line with storage buffers, what does it mean if a buffer is nearly always empty or nearly always full?
-
Carbon-14 dating is used to date a bone found at an archaeological excavation. If the ratio of C-14 to C-12 atoms is 3.25 1013, how old is the bone?
-
A sample of radioactive 21483Bi, which has a half-life of 19.9 min, has an activity of 0.058 Ci. What is its activity 1.0 h later?
-
The activity of a sample containing radioactive 108Ag is 6.4 104 Bq. Exactly 12 min later, the activity is 2.0 103 Bq. Calculate the half-life of 108Ag.
-
Find the first derivative of the following equation: a. b. y=e3x y=e(x+1) d. y=125x+16-2x y=ln x y=ln (3x+x1) y=In (x+1)
-
The Cutting Department of Lasso Company has the following production and cost data for August. Production Costs 1. Started and completed 9,600 units. Beginning work in process $0 2. Started 2,300...
-
There is sufficient evidence to support the conclusion that a robust relationship exists between drugs and crime. Explain and discuss the studies that support this conclusion. Further explain and...

Study smarter with the SolutionInn App


