Explain why, for these three substances, the solubility in 20C water goes down as the molecules get
Question:
Explain why, for these three substances, the solubility in 20°C water goes down as the molecules get larger, but the boiling point goes up.
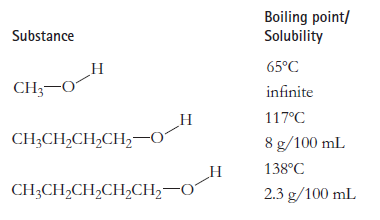
Transcribed Image Text:
Boiling point/ Solubility Substance 65°C Н CH;-0 infinite 117°C Н CH;CH,CH,CH,–0 8 g/100 mL 138°C CH3CH,CH,CH,CH2–0 2.3 g/100 mL
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
The boiling points go up because of an increase in the number of molecular interactions between mol...View the full answer

Answered By

Abhitansh GUPTA
I am having a one year of dedicated experience of subject Matter expert in Indian based company so following international codes is not an issue along with I am having good understanding of the type of problems asked in various overseas universities.
Fetching you good grades is my responsibility don't worry about that.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Conceptual Physical Science
ISBN: 978-0134060491
6th edition
Authors: Paul G. Hewitt, John A. Suchocki, Leslie A. Hewitt
Question Posted:
Students also viewed these Physics questions
-
The solubility of nitrogen in water is 8.21 10-4 mol/ L at 0oC when the N2 pressure above water is 0.790 atm. Calculate the Henrys law constant for N2 in units of L atm/mol for Henrys law in the...
-
The solubility of carbon dioxide in water is 0.161 g CO2 in 100 mL of water at 20C and 1.00 atm. A soft drink is carbonated with carbon dioxide gas at 5.50 atm pressure. What is the solubility of...
-
Explain why pressure substantially affects the solubility of O2 in water but has little effect on the solubility of NaCl in water?
-
(a) Write an equation describing a sinusoidal transverse wave traveling on a cord in the positive direction of a y axis with an angular wave number of 60 cm-1, a period of 0.20 s, and an amplitude of...
-
Fletcher Corporation has hired a marketing representative to sell the company's two products: Marvelous and Wonderful. The representative's total salary and fringe benefits are $8,000 monthly. The...
-
Determine the horizontal and vertical components of reaction at the supports A and B. Assume the joints at C and D are fixed connections. 40 kN 30 kN 20 kN 12 kN/m 4 m B - 6 m- - 8 m
-
Derive an expression for the natural frequencies for the lateral vibration of a uniform fixedfree beam.
-
The Svenson Corporation manufactures cellular modems. It manufactures its own cellular modem circuit boards (CMCB), an important part of the cellular modem. It reports the following cost information...
-
During the current fiscal year, Jeremiah Corp. signed a long-term non-cancellable purchase commitment with its primary supplier. Jeremiah agreed to purchase $2,580,000 of raw materials during the...
-
1. Whats your reaction to these events? Do you think your reaction is influenced by the culture, values, and traditions of the country in which you find yourself? Explain. 2. Look at what Hofstedes...
-
Which should weigh more: 100 mL of fresh water or 100 mL of fresh sparkling seltzer water? Why? Which should weigh more: 100 mL of flat seltzer water at 20C or the same 100 mL of flat seltzer water...
-
The boiling point of 1,4-butanediol is 230C. Would you expect this compound to be soluble or insoluble in room-temperature water? Explain. -,CH,CH,CH, 0 1,4-Butanediol
-
Refer to the Journal of Food Engineering (Sep. 2013) study of the characteristics of fried sweet potato chips, Exercise 7.78. Recall that a sample of 6 sweet potato slices fried at 130 using a vacuum...
-
How can income statement data be used to estimate cash flow?
-
In the Ricardian trade model, comparative advantage is determined by: A. technology. B. the capital-to-labor ratio. C. the level of labor productivity.
-
What are the two primary tests that good cost allocation processes pass?
-
Describe the ways a salesperson is able to add value through customer follow-up.
-
How does the statement differ between not-for-profit and forprofit entities?
-
A 2013 survey by the Pew Research Center asked whether there have been times in the past year the respondent has been unable to afford food. Of advanced economies, the country with the second highest...
-
You have accepted the engagement of auditing the financial statements of the C. Reis Company, a small manufacturing firm that has been your auditee for several years. Because you were busy writing...
-
The black dots at the top of Fig. Q2.2 represent a series of high-speed photographs of an insect flying in a straight line from left to right (in the positive x-direction). Which of the graphs in...
-
Can an object with constant acceleration reverse its direction of travel? Can it reverse its direction twice? In both cases, explain your reasoning.
-
Under what conditions is average velocity equal to instantaneous velocity?
-
Athens Gas Station has figured out the weekly demand distribution for their gas sales. Each gallon of gas sold at the pump results in a profit of 10 cents/gallon and any lost sales results in a cost...
-
1. The maximum pressure an eardrum can withstand without rupturing is Pmax = 3.0102 Pa. Assume a sound is made at that pressure. If the density of air is, p=1.20 kg/m, and the speed of sound is 340...
-
52. A wave is sent down a string with a linear mass density of 1.600 x 10-4 kg/m. The wavefunction that describes this wave is: y(x,t) = (0.021 m)*sin{(2.00 rad/m)*x + (30.0 rad/s)*t} What is the...

Study smarter with the SolutionInn App


