Place the proper number of electrons in each shell: Rubidium, Rb Sodium, Na Krypton, Kr Chlorine, CI
Question:
Place the proper number of electrons in each shell: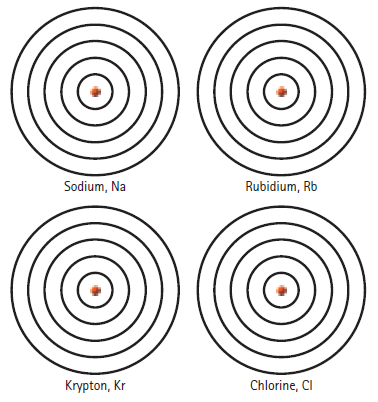
Transcribed Image Text:
Rubidium, Rb Sodium, Na Krypton, Kr Chlorine, CI
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
Sodium Na ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Conceptual Physical Science
ISBN: 978-0134060491
6th edition
Authors: Paul G. Hewitt, John A. Suchocki, Leslie A. Hewitt
Question Posted:
Students also viewed these Physics questions
-
For germanium (Ge, Z = 32), make a list of the number of electrons in each sub shell (1s, 2s, 2p, . .). Use the allowed values of the quantum numbers along with the exclusion principle; do not refer...
-
Complete the molecular orbital description for the ground state of cyclopentadiene shown at right. Shade the appropriate lobes to indicate phase signs in each molecular orbital according to...
-
What is the maximum number of electrons in the entire n = 2 shell?
-
Barry consumes tacos and pepsi. The price of tacos is initially $4.00 per taco and the price of pepsi is $1.00 per can and Bary has $20.00 to spend. The marginal utility that each good yields is...
-
A company wants to raise $500 million in a new stock issue. Its investment banker indicates that the sale of new stock will require 8 percent under pricing and a 7 percent spread. (Hint: the...
-
Consider a version of the pipeline from Section 4.6 that does not handle data hazards (i.e., the programmer is responsible for addressing data hazards by inserting NOP instructions where necessary)....
-
In a survey, U.S. adults were asked to identify which social media platforms they use. The results are shown in the figure. Six adults who participated in the survey are randomly selected and asked...
-
If you buy a callable bond and interest rates decline, will the value of your bond rise by as much as it would have risen if the bond had not been callable? Explain.
-
Because of its high dielectric strength, SF6 (sulfur hexafluo-ride) gas is widely used as an insulator and a dielectric in HV applications such as HV transform-ers, switches, circuit breakers,...
-
Given the information provided in the chapter, discuss the psychological phenomena associated with Martha Stewarts investment decisions.
-
Some older cars vibrate loudly when driving at particular speeds. For example, at 65 mph the car may be most quiet, but at 60 mph the car may rattle uncomfortably. How is this analogous to the...
-
Use the shell model to explain why a potassium atom, K, is larger than a sodium atom, Na.
-
On March 25, Hannah received an invoice in the mail from Carpet Country for $4235. The invoiced was dated March 22 and had terms 3/15, 1 /30, n/60. If she made payments of $1000 on each of April 2,...
-
What are the reasons why the once - popular PL/I - derivative languages, such as Intel s PL/M, Motorola s MPL and Zilog s PL/Z, practically disappeared by the late 1980s (see Fig. 4.1 )? Figure...
-
Estimate and justify the relative percentage of person months spent in each phase of the requirements engineering process (see Figure 5.1 ) for some embedded real - time system. The instructor is...
-
Use standard compiler - optimization methods and multiple optimization phases to optimize the following C code by hand: #define UNIT 1 #define FULL 1 void main(void) { int a,b; a=FULL; b=a; if...
-
The door - open button in Figure 5.2 is of momentary - pressure type. Hence, it is enough to press the button momentarily to open the door. However, when there is a fi re in the building and the...
-
Find at least three advantages and three disadvantages of the HSMC architecture (see Fig. 9.2 ). Based on those findings, evaluate your confidence (scale 0 1) on this particular hardware vision. By...
-
The following stock quotations were recently reported in The Wall Street Journal: a. What are the dividend yields on the common stock of AT&T, Boeing, and Johnson & Johnson? b. What possible...
-
2.) Find the Laplace transform of f(t) 7e-St cos 2t +9 sinh2 2t. Use Laplace Table. %3D
-
A current sheet, K = 20 a z A/m, is located at = 2, and a second sheet, K = 10a z A/m, is located at = 4. (a) Let V m = 0 at P( = 3, = 0, z = 5) and place a barrier at = . Find V m (, , z) for <
-
Assume that A = 50 2 a z Wb/m in a certain region of free space. (a) Find H and B. (b) Find J. (c) Use J to find the total current crossing the surface 0 1, 0 < 2, z = 0. (d) Use the value of H ...
-
Show that 2 (1/R 12 ) = 1 (1/R 12 ) = R 21 /R 3 12 .
-
(6 pts.) The following gas phase reaction system is conducted in an isothermal constant volume batch reactor. The reactor is charged with pure A with an initial concentration CAO AB ri = k CA B D r2...
-
Based on the NPV and IRR investment criteria, should your company introduce a new candy flavor? 1) Should the cost of a marketing survey be included in your decision? Why or why not? 2) Calculate the...
-
You invest into Malaysian stock market. On 3 May 2021, construct an investment portfolio. State the reason(s) for selecting those financial assets available in the Malaysian stock market. (b) To...

Study smarter with the SolutionInn App


