What must be added to a double bond to transform it into an alcohol? Would this be
Question:
What must be added to a double bond to transform it into an alcohol? Would this be an example of oxidation or reduction? (See Figure 18.20)
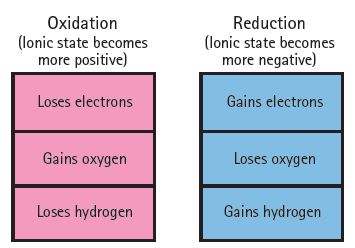
Transcribed Image Text:
Oxidation (lonic state becomes more positive) Reduction (lonic state becomes more negative) Loses electrons Gains electrons Gains oxygen Loses oxygen Gains hydrogen Loses hydrogen
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)
Adding a water molecule to ...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Conceptual Physical Science
ISBN: 978-0134060491
6th edition
Authors: Paul G. Hewitt, John A. Suchocki, Leslie A. Hewitt
Question Posted:
Students also viewed these Physics questions
-
What are the six FRs of a stable manufacturing system? a. W. Edwards Deming said that to know when a problem exists we must distinguish normal from abnormal. How and why does standardized work make...
-
What volume of 0.0100 M NaOH must be added to 1.00 L of 0.0500 M HOCl to achieve a pH of 8.00?
-
What mass of steam at 100oC must be added to 1.00 kg of ice at 0oC to yield liquid water at 20oC?
-
Data Corporation has four employees and provides group term life insurance coverage for all four employees. Coverage is nondiscriminatory and is as follows: a. How much may Data Corporation deduct...
-
Erin attends graduate school to earn a master's degree in accounting. Her parents, the Martins, pay Erin's tuition for the program. Early in 2014, Erin's dad left his job and took a few months off to...
-
A commercial bank made a five-year term loan at 13 percent. The bank's economics department forecasts that one and three years in the future, the two-year interest rate will be 12 percent and 14...
-
Write an equation for calculating the cost of savings life cycle economics of a proposed passive solar system. Explain why it is important to be able to determine the auxiliary energy required for...
-
This problem continues the Daniels Consulting situation from Problem P2-41 of Chapter 2. You will need to use the unadjusted trial balance and posted T-accounts that you prepared in Problem P2-41....
-
A large positively charged object with charge 9+ = 4.75 C is brought near a negatively charged plastic ball suspended from a string of negligible mass. The suspended ball has a charge of q = -48.3 nC...
-
Campus Theater adjusts its accounts every month. The company's unadjusted trial balance dated August 31, current year, appears as follows. Additional information is provided for use in preparing the...
-
Identify the following functional groups in this organic molecule: amide, ester, ketone, ether, alcohol, aldehyde, amine.
-
What is the difference between a ketone and an aldehyde?
-
Why are some industries more global than others? Discuss.
-
What is a manufacturing network?
-
The value of a payment depends on when it is made. a. Future value is the present value of an initial investment times one plus the interest rate for each year you hold it. The higher the interest...
-
Explain the role of the customer in a total quality setting.
-
Give a brief rationale for empowerment.
-
Jane and Mike purchase identical houses for $400,000. Jane makes a down payment of $80,000, while Mike puts down only $20,000; for each individual, the down payment is the total of his or her net...
-
Find the general solution of the given differential equation. Give the largest interval I over which the general solution is defined. Determine whether there are any transient terms in the general...
-
A sample statistic will not change from sample to sample. Determine whether the statement is true or false. If it is false, rewrite it as a true statement.
-
A horizontal spring with spring constant 85 N/m extends outward from a wall just above floor level. A 1.5 kg box sliding across a frictionless floor hits the end of the spring and compresses it 6.5...
-
One mole (6.02 10 23 atoms) of helium atoms in the gas phase has 3700 J of microscopic kinetic energy at room temperature. If we assume that all atoms move with the same speed, what is that speed?...
-
A 55 kg softball player slides into second base, generating 950 J of thermal energy in her legs and the ground. How fast was she running?
-
a) The function is increasing. The table below gives values for the function at selected values of x. A logarithmic regression y=a+blnx is used to model the data. Find the logarithmic regression...
-
4. It can be shown using material from a later section that is given by 0-122 dx (-1)na2n+1 n!2n (2n+1) n=0 for any positive value of a. When a is positive, this series is an alternating series. (a)...
-
Using desmos, graph each of the following functions. Identify the open interval(s) for which the requested characteristic is displayed. #1 ON [-2,2] FUNCTION INCREASING f(x)=xcosx (0.96,0.96) FIRST...

Study smarter with the SolutionInn App


