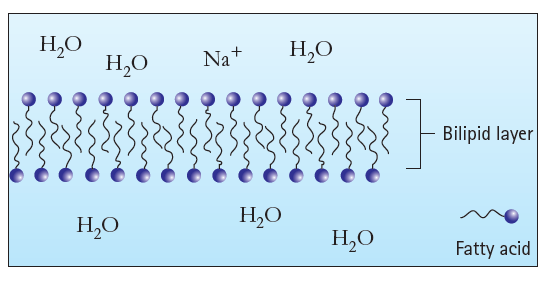
Why do nonpolar molecules have a difficult time passing through the bilipid layer? How about polar molecules?
Question:
Why do nonpolar molecules have a difficult time passing through the bilipid layer? How about polar molecules?
Transcribed Image Text:
Н.о НО Na+ Н,О Bilipid layer Н.О НО Но Fatty acid
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
The nonpolar molecules have a hard time passing the ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Conceptual Physical Science
ISBN: 978-0134060491
6th edition
Authors: Paul G. Hewitt, John A. Suchocki, Leslie A. Hewitt
Question Posted:
Students also viewed these Physics questions
-
The three chemicals listed below are all very weak acids because they all have a difficult time losing a hydrogen ion, H + . Upon losing this hydrogen ion, the central atom of each of these molecules...
-
(a) Find the mean kinetic energy in a of molecules that exits from a small hole in an oven at temperature . (b) Assume now that the molecules are collimated by a second hole farther down the beam, so...
-
In this problem you are to find how the van der Waals force between a polar and a nonpolar molecule depends on the distance between the molecules. Let the dipole moment of the polar molecule be in...
-
Spherical refracting surfaces an object O stands on the central axis of a spherical refracting surface. For this situation, each problem in Table 34-5 refers to the index of refraction n1 where the...
-
Production workers for Chadwick Manufacturing Company provided 3,200 hours of labor in January and 2,800 hours in February. The company, whose operation is labor intensive, expects to use 48,000...
-
Joan Bennish began Bennish Consulting on May 1, 2017, and reported the items below at May 31, 2017. Match each numbered item with the part of the balance sheet on which it should be presented. If the...
-
Fill in the Blank. The ____________ energy of a beam is denoted by \(\frac{1}{2} \int_{0}^{l} E I\left(\frac{\partial^{2} w}{\partial x^{2}} ight)^{2} d x\).
-
Trudy Company incurred the following costs. 1. Sales tax on factory machinery purchased $5,000 2. Painting of and lettering on truck immediately upon purchase 700 3. Installation and testing of...
-
A 1000 gallon holding tank that catches runoff from some chemical process initially has 800 gallons of water with 2 ounces of pollution dissolved in it. Polluted water flows into the tank at a rate...
-
Monochromatic light is beamed into a Michelson interferometer. The movable mirror is displaced 0.382 mm, causing the interferometer pattern to reproduce itself 1 700 times. Determine the wavelength...
-
Which of these boxes best represents a suspension?
-
Why is flushing a toilet with clean water from a municipal supply about as wasteful as flushing it with bottled water?
-
In Exercises 21 through 36, solve the given equation for x. ln x = 2(ln 3 ln 5)
-
Briefly explain why the base case analysis required the calculation to move up the P&L statement rather than down (the normal direction).
-
Why is the number of members so important in a capitated environment?
-
Define the term relevant range.
-
What is meant by the term cost?
-
Why is utilization management so important in a capitated environment?
-
Consider the test of H 0 : The defendant is not guilty against H a : The defendant is guilty. a. Explain, in context, the conclusion of the test if H 0 is rejected. b. Describe, in context, a Type I...
-
Why is disclosure of depreciation or amortization methods and rates so important?
-
A soap bubble is essentially a very thin film of water (n = 1.33) surrounded by air. The colors that you see in soap bubbles are produced by interference. a. Derive an expression for the wavelengths ...
-
A sheet of glass is coated with a 500-nm-thick layer of oil (n = 1.42). a. For what visible wavelengths of light do the reflected waves interfere constructively? b. For what visible wavelengths of...
-
Two loudspeakers emit sound waves along the x-axis. A listener in front of both speakers hears a maximum sound intensity when speaker 2 is at the origin and speaker 1 is at x = 0.50 m. If speaker 1...
-
Evaluate economic conditions that influence company performance of starbucks Consider political, environmental, currency ( money ) , global economics, and government influences on economic...
-
(Part 1) : The business letter to your employer. The purpose of the letter is to explain why diversity is important in the workplace and to recommend actions the organization can take to become more...
-
Question 4 (20 marks)In the recent annual general meeting of Umi Technology Limited, the board of directors declared an annual dividend of $5.00 per share, and the shareholders agreed to a rights...

Study smarter with the SolutionInn App


