Toot Auto Supply distributes new and used automobile parts to local dealers throughout the Midwest. Toots credit
Question:
Toot Auto Supply distributes new and used automobile parts to local dealers throughout the Midwest. Toot’s credit terms are n/30. As of the end of business on October 31, the following accounts receivable were past due:
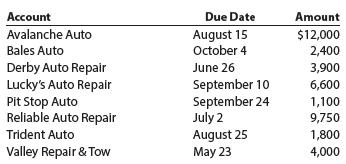
Determine the number of days each account is past due as of October 31.
Transcribed Image Text:
Account Due Date Amount August 15 October 4 Avalanche Auto $12,000 2,400 Bales Auto Derby Auto Repair Lucky's Auto Repair Pit Stop Auto Reliable Auto Repair June 26 3,900 September 10 September 24 July 2 August 25 May 23 6,600 1,100 9,750 Trident Auto 1,800 Valley Repair & Tow 4,000
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
Account Receivables It is the amount to be received within a specific shorter period from customers ...View the full answer

Answered By

Vipul Bhalara
I have a total of 6 years of experience in Accounting and Finance subjects with the various online portal.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Corporate Financial Accounting
ISBN: 9781337398169
15th Edition
Authors: Carl S. Warren, James M. Reeve, Jonathan Duchac
Question Posted:
Students also viewed these Business questions
-
Bubba's Auto Supply distributes new and used automobile parts to local dealers throughout the Southeast. Bubba's credit terms are n/30. As of the end of business on July 31, the following accounts...
-
Honest Abes Auto Supply distributes new and used automobile parts to local dealers throughout the Northeast. Honest Abes credit terms are n/30. As of the end of business on July 31, the following...
-
On 30 October the following were among the balances in the cost ledger of a company manufacturing a single product (Product X) in a single process operation: The raw material ledger comprised the...
-
A product has a contribution margin of $6 per unit and a selling price of $30 per unit. Fixed costs are $24,000. Assuming the new technology increases the unit contribution margin by 60 percent but...
-
With blending inheritance, the height of the offspring is equal to the average height of the four grandparents. Find the probability that all four grandparents have height 40 cm and thus the...
-
Carol feels that the best way to determine how much life insurance she needs is to use the capital drawdown method. If inflation is 2.1% and she wants to have enough life insurance to provide her...
-
For the similarity solution, what are the boundary conditions for the constant-wall-flux case? Show that a complete similarity does not exist for this case. Also show the condition for the case where...
-
On June 30, 2018, a county (government) hospital bought 3,000 shares of stock for $94,000 intending to hold the investment for its proposed expansion of its trauma center. The market value of the...
-
On January 1, 2023, Stream Company acquired 25 percent of the outstanding voting shares of Q-Video, Incorporated, for $718,000. Q- Video manufactures specialty cables for computer monitors. On that...
-
(a) Prepare the following consolidated financial statements for Year 6: (i) Income statement (ii) Statement of financial position (b) Calculate goodwill impairment loss and profit attributable to...
-
The following data were accumulated for use in reconciling the bank account of Creative Design Co. for August 20Y6: 1. Cash balance according to the companys records at August 31, $42,920. 2. Cash...
-
Using the data in Exercise 8-11, assume that the allowance for doubtful accounts for Outlaw Bike Co. had a debit balance of $5,140 as of December 31. Journalize the adjusting entry for uncollectible...
-
A client seeking liquidity sets aside 35,000 in a bank account today. The account pays 5 percent compounded monthly. Because the client is concerned about the fact that deposit insurance covers the...
-
A certain container holds 50 monatomic particles with a combined energy of 200 units. 25 diatomic particles, with each particle having 2 units of energy, str then added and the system is allowed to...
-
Find the rate of change of entropy with respect to thermal energy of a system with an absolute temperature of \(100{ }^{\circ} \mathrm{C}\).
-
You poured freshly brewed hot tea into a cup. A thermometer indicates that its temperature is \(96.8^{\circ} \mathrm{C}\). Convert this temperature into the Fahrenheit and Kelvin scales.
-
Please arrange the following five symbols into an equation with no minus signs in it. A1 = assets at end of period. L1 = liabilities at end of period. OE0 = owners equity at beginning of period. R1 =...
-
Three monatomic ideal gas particles have the following velocities (written in component form) in a coordinate system: \((3.0,4.0) \mathrm{m} / \mathrm{s},(-6.0,8.0) \mathrm{m} /...
-
The electromagnetic wave that delivers a cellular phone call to a car has a magnetic field with an rms value of 1.5 10-10 T. The wave passes perpendicularly through an open window, the area of which...
-
Use this circle graph to answer following Exercises. 1. What fraction of areas maintained by the National Park Service are designated as National Recreation Areas? 2. What fraction of areas...
-
Wilmington Chemical Company produces three products: ethylene, butane, and ester. Each of these products has high demand in the market, and Wilmington Chemical is able to sell as much as it can...
-
Wilmington Chemical Company produces three products: ethylene, butane, and ester. Each of these products has high demand in the market, and Wilmington Chemical is able to sell as much as it can...
-
Wilmington Chemical Company produces three products: ethylene, butane, and ester. Each of these products has high demand in the market, and Wilmington Chemical is able to sell as much as it can...
-
1. Mention five key organizational characteristics that the Cll research team identified for organizations with effective quality management systems. provide brief discussion of each and how it...
-
When a metal was exposed to photons at a frequency of 1.46 1015 s1, electrons were emitted with a maximum kinetic energy 3.60 10-19 J. G ? Calculate the work function, , of this metal. J/photon What...
-
No. 101 Debit Cash $ 2,400 Question 2 (10 pts) On November 1, 2024, the account balances of Schilling Equipment Repair were as follows. Accumulated Depreciation-Equipment A Clipboard Font Alignment...

Study smarter with the SolutionInn App


