Consider the split-split-plot design described in Example 13-3. Suppose that this experiment is conducted as described and
Question:
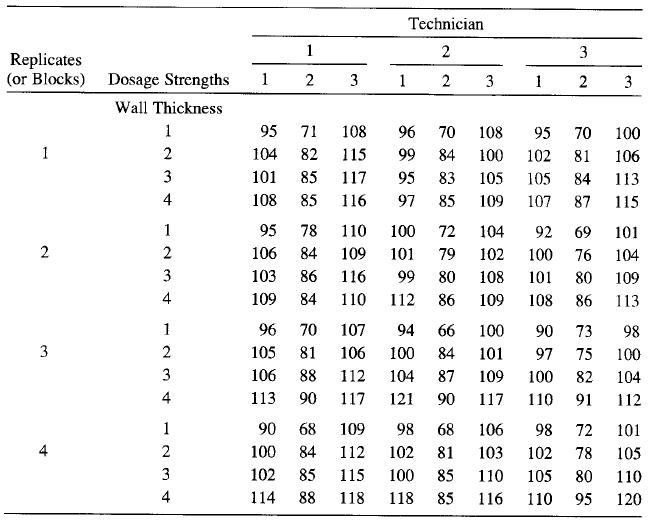
Consider the split-split-plot design described in Example 13-3. Suppose that this experiment is conducted as described and that the data shown in the following table are obtained. Analyze the data and draw conclusions.
Transcribed Image Text:
Technician 1 2 3 Replicates (or Blocks) Dosage Strengths 1 1 3 1 3 Wall Thickness 1 95 71 108 96 70 108 95 70 100 1 104 82 115 99 84 100 102 81 106 101 85 117 95 83 105 105 84 113 4 108 85 116 97 85 109 107 87 115 1 95 78 110 100 72 104 92 69 101 2 2 106 84 109 101 79 102 100 76 104 3 103 86 116 99 80 108 101 80 109 4 109 84 110 112 86 109 108 86 113 1 96 70 107 94 66 100 90 73 98 2 105 81 106 100 84 101 97 75 100 3 106 88 112 104 87 109 100 82 104 4 113 90 117 121 90 117 110 91 112 1 90 68 109 98 68 106 98 72 101 2 100 84 112 102 81 103 102 78 105 3 102 85 115 100 85 110 105 80 110 4 114 88 118 118 85 116 110 95 120 3. 2. 434 3. 4.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
In this splitsplitplot design the wall thicknesses were measured for different technicians replicate...View the full answer

Answered By

SHINKI JALHOTRA
I have worked with other sites like Course Hero as a tutor and I have great knowledge on IT skills.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Consider a repeated version of the game in exercise 24.5. In this version, we do not give all the proposal power to one person but rather imagine that the players are bargaining by making different...
-
Rework Problem 13-22, assuming that the technicians are chosen at random. Use the restricted form of the mixed model. Problem 13-22 Consider the split-split-plot design described in Example 13-3....
-
Consider the situation described in Example 3.7.14. Suppose that X1 = 5 and X2 = 7 are observed. a. Compute the conditional p.d.f. of X3 given (X1, X2) = (5, 7). (You may use the result stated in...
-
2. Two point charges are located at two comers of a triangle as shown. What is the electric potential at the right corner of the triangle? A. 21 10 V C. 4.5 x 10 V B. 3.4 x 10 V D. 6.3 x 10 V 10 em...
-
For each of the following discrete-time dynamical systems, indicate which of the equilibria are stable and which are unstable. Plants 1000 r 800 600 400 200 0 200 400 600 800 1000 Initial population
-
Consider the following cash flows and present-worth profiles on Page 218: (a) Determine the values for X and Y. (b) Calculate the terminal project balance of Project 1 at MARR = 24%. (c) Find the...
-
With reference to Exercise 10.1, what can we say with \(95 \%\) confidence about the maximum error if we use the sample proportion as an estimate of the true proportion of complaints filed against...
-
Your division is considering two investment projects, each of which requires an up-front expenditure of $15 million. You estimate that the investments will produce the following net cash flows: What...
-
What are three resource management tools that facilitate family decision making? Discuss how to structure a successful performance appraisal interview?
-
Refer to the feasible region for Par, Inc., in Problem 14. a. Develop an objective function that will make extreme point (0, 540) the optimal extreme point. b. What is the optimal solution for the...
-
Repeat Problem 13-20, assuming that the mixes are random and the application methods are fixed. Problem 13-20 An experiment is designed to study pigment dispersion in paint. Four different mixes of a...
-
Reconsider the experiment in Problem 5-22. Use the Box-Cox procedure to determine if a transformation on the response is appropriate (or useful) in the analysis of the data from this experiment....
-
The oxygen consumed during the oxidation of glucose in animal cells is returned as CO 2 to the atmosphere. Is the statement true? Explain why or why not.
-
Jan 5: Purchased land, equipment, and a building. The land cost $1,000,000, the equipment cost $250,000, and the building cost $1,000,000. The CEO said the land depreciates via the Straight-line...
-
Vicki teaches accounting courses at a community college. The college administration suspects that at least a few employees are improperly using their computers. Hidden cameras are installed around...
-
Benjamin's Production Company has recently suffered a large theft of production equipment during the filming of a movie. The theft was only discovered the next morning when he noticed that the...
-
On February 1 , 2 0 2 0 , the Greece Corp., which maintains a perpetual inventory system, acquired merchandise inventory by signing a two - year, 1 4 % , $ 8 0 , 0 0 0 note payable. On this date, the...
-
Snow resorts are highly dependent on seasonal weather and suffer from low visitations during off-ski seasons. Discuss strategies on how ski resorts can manage demand during non-snow seasons. What...
-
Why is it virtually impossible to measure whether a process is capable (in the sense of having a high probability of being within specification limits) if it isn't in control?
-
B.) What is the approximate concentration of free Zn 2+ ion at equilibrium when 1.0010 -2 mol zinc nitrate is added to 1.00 L of a solution that is 1.080 M in OH - . For [Zn(OH) 4 ] 2- , K f = 4.610...
-
Use MuPAD to determine all the local minima and local maxima and all the inflection points where dy/dx = 0 of the following function: y = x - + 8x - 4 3
-
The surface area of a sphere of radius r is S = 4r 2 . Its volume is V = 4r 3 /3. a. Use MuPAD to find the expression for dS/dV. b. A spherical balloon expands as air is pumped into it. What is the...
-
Use MuPAD to find the point on the line y = 2 x/3 that is closest to the point x = -3, y = 1.
-
You invest into Malaysian stock market. On 3 May 2021, construct an investment portfolio. State the reason(s) for selecting those financial assets available in the Malaysian stock market. (b) To...
-
Line spectra for atoms is generated by electronic transitions between quantum energy levels. For the hydrogen atom we established that in the absence of external fields the energy depends only on the...
-
Control Substances: 3.0 g calcium carbonate (chalk) and 0.50 M HCI CaCO3(s) + 2HCl(aq) CaCl2(s) + H2O(aq) + CO2(g) Can you please 2 graphs, one for the control experiment and one for the variable...

Study smarter with the SolutionInn App


