Four chemists are asked to determine the percentage of methyl alcohol in a certain chemical compound. Each
Question:
Four chemists are asked to determine the percentage of methyl alcohol in a certain chemical compound. Each chemist makes three determinations, and the results are the following:
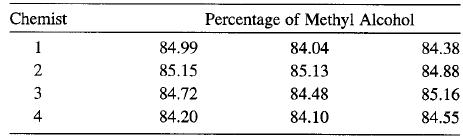
(a) Do chemists differ significantly? Use α = 0.05.
(b) Analyze the residuals from this experiment.
(c) If chemist 2 is a new employee, construct a meaningful set of orthogonal contrasts that might have been useful at the start of the experiment.
Transcribed Image Text:
Chemist Percentage of Methyl Alcohol 1 84.99 84.04 84.38 85.15 85.13 84.88 3 84.72 84.48 85.16 4 84.20 84.10 84.55
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
ANSWER To determine if the chemists differ significantly in their measurements of the percentage of methyl alcohol we can perform an analysis of variance ANOVA test a Hypothesis testing Null hypothesi...View the full answer

Answered By

Surendar Kumaradevan
I have worked with both teachers and students to offer specialized help with everything from grammar and vocabulary to challenging problem-solving in a range of academic disciplines. For each student's specific needs, I can offer explanations, examples, and practice tasks that will help them better understand complex ideas and develop their skills.
I employ a range of techniques and resources in my engaged, interesting tutoring sessions to keep students motivated and on task. I have the tools necessary to offer students the support and direction they require in order to achieve, whether they need assistance with their homework, test preparation, or simply want to hone their skills in a particular subject area.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Four laboratories (1-4) are randomly selected from a large population, and each is asked to make three determinations of the percentage of methyl alcohol in specimens of a compound taken from a...
-
You've been asked to determine the percentage of students who would support gay marriage. You want to take a random sample of fellow students to make the estimate. Explain whether each of the...
-
Social networking is becoming more and more popular around the world. Pew Research Center used a survey of adults in several countries to determine the percentage of adults who use social networking...
-
Consider a tank containing a liquid, and the rate of change of the liquid's height (h) with respect to time (t) is proportional to the difference between the current height and a reference height....
-
Sketch graphs of the following updating function over the given range and mark the equilibria. h(z) = e-z for 0 z 2.
-
Water at 10C ( = 999.7 kg/m 3 and = 1.307 10 -3 kg/ms) is flowing steadily in a 0.12-cm-diameter, 15-m-long pipe at an average velocity of 0.9 m/s. Determine (a) The pressure drop, (b) The head...
-
The front door of a dishwasher of width \(580 \mathrm{~mm}\) has a vertical air vent that is \(500 \mathrm{~mm}\) in height with a \(20-\mathrm{mm}\) spacing between the inner tub operating at...
-
Natalie decides that she cannot afford to hire John to do her accounting. One way that she can ensure that her cash account does not have any errors and is accurate and up-to-date is to prepare a...
-
Discuss the role of bifunctional catalysis in asymmetric synthesis. How does the cooperative action of multiple catalytic sites within a single catalyst molecule contribute to enhanced stereocontrol ?
-
Air FranceKLM (AF), a Franco-Dutch company, prepares its financial statements according to International Financial Reporting Standards. AFs financial statements and disclosure notes for the year...
-
The effective life of insulating fluids at an accelerated load of 35 kV is being studied. Test data have been obtained for four types of fluids. The results were as follows: (a) Is there any...
-
Three brands of batteries are under study. It is suspected that the lives (in weeks) of the three brands are different. Five batteries of each brand are tested with the following results: (a) Are the...
-
What type of legal compliance strategy would you advise an employer to adopt? Why? Pushing the envelope in areas of legal uncertainty or erring on the the side of caution? Doing no more than the...
-
A crate rests on a horizontal surface and a woman pulls on it with a 10-N force. No matter what the orientation of the force, the crate does not move. Rank the situations below according to the...
-
Ralph Henderson owns a cafe near MIT. His cafe is always crowded with students who need caffeine to start their day. Ralph Henderson wants to serve the freshest coffee to his customers. He would like...
-
The single particle energy eigenstates of a hypothetical system are nondegenerate and have energies given by a = o: where & = 6.25 x 10-21 eV. The index a runs over the positive integers. The system...
-
-3 A thermally conductive rod of cross sectional area 7.52 x 10 m2, length 0.78 m, and conductivity 345 W/mK, connects two containers. In the left container is a very large quantity of a substance at...
-
Trevor Mills produces agricultural feed at its only plant. Materials are added at the beginning of the process. Information on work - in - process in December follows: Beginning inventory, 5 6 , 0 0...
-
How is an ERP system built around the concept of a central repository for information?
-
The tractor is used to lift the 150-kg load B with the 24-mlong rope, boom, and pulley system. If the tractor travels to the right at a constant speed of 4 m/s, determine the tension in the rope when...
-
Write the charge balance for a solution of H 2 SO 4 in water if H 2 SO 4 ionizes to HSO 4 and SO 2 4 - .
-
For a 0.1 M aqueous solution of sodium acetate, Na + CH 3 CO - 2 , one mass balance is simply [Na + ] = 0.1 M. Write a mass balance involving acetate.
-
(a) Following the example of Mg(OH) 2 in Section 7-5, write the equations needed to find the solubility of Ca(OH) 2 . Include activity coefficients where appropriate. Equilibrium constants are in...
-
Create a class named Account that contains: A private int data field named id for the account (default 0). A private double data field named balance for the account (default 0). A private double data...
-
The next Java code needs to be converted from static to dynamic (just addd changes in same code): import java.util.Scanner; class MyIntStaticCircularQueue { int capacity = 2; int queue[] = new...
-
Hi! Would a tutor be able to assist me on this? In a complete graph with 48 vertices, how many vertices will be in each node's adjacency list? How many entries will be in the adjacency matrix for...

Study smarter with the SolutionInn App


