Use the modified Levene test to determine if the assumption of equal variances is satisfied in Problem
Question:
Use the modified Levene test to determine if the assumption of equal variances is satisfied in Problem 3-14. Use α = 0.05. Did you reach the same conclusion regarding the equality of variances by examining residual plots?
Problem 3-14.
Three brands of batteries are under study. It is suspected that the lives (in weeks) of the three brands are different. Five batteries of each brand are tested with the following results:
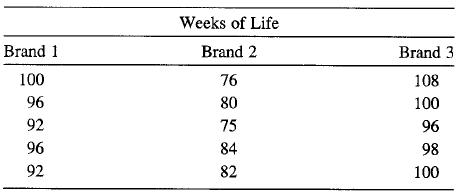
Transcribed Image Text:
Weeks of Life Brand 1 Brand 2 Brand 3 100 76 108 96 80 100 92 75 96 96 84 98 92 82 100
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 81% (11 reviews)
o determine if the assumption of equal variances is satisfied in Problem 314 we can use the modified Levene test The modified Levene test is a statist...View the full answer

Answered By

Churchil Mino
I have been a tutor for 2 years and have experience working with students of all ages and abilities. I am comfortable working with students one-on-one or in small groups, and am able to adapt my teaching style to meet the needs of each individual. I am patient and supportive, and my goal is to help my students succeed.
I have a strong background in math and science, and have tutored students in these subjects at all levels, from elementary school to college. I have also helped students prepare for standardized tests such as the SAT and ACT. In addition to academic tutoring, I have also worked as a swim coach and a camp counselor, and have experience working with children with special needs.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Use Bartlett's test to determine if the assumption of equal variances is satisfied in Problem 3-14. Use = 0.05. Did you reach the same conclusion regarding equality of variances by examining...
-
The paper The Truth About Lying in Online Dating Profiles (Proceedings, Computer-Human Interactions [2007]: 14) describes an investigation in which 40 men and 40 women with online dating profiles...
-
Use software to conduct the log-rank test to determine if the melting experiences for the white and milk chocolate chips in your chip melting activity are different. What do you conclude about the...
-
Necked Amber purchased a bond for $1,038.90 exactly two years ago. At that time, the bond had a maturity of five years and a coupon rate of 10% (paid semi-annually). Assuming the rates below are the...
-
Graph the updating functions associated with the following discrete-time dynamical systems, and cobweb for five steps starting from the given initial condition. wt+1 = -0.5wt + 3, starting from w0 =...
-
In preparation for a proposed bond sale, the city manager of the City of Appleton requested that you prepare a statement of legal debt margin and a schedule of direct and overlapping debt for the...
-
Consider a horizontal 5-mm-thick, 100-mm-long straight fin fabricated from plain carbon steel \((k=57\) \(\mathrm{W} / \mathrm{m} \cdot \mathrm{K}, \varepsilon=0.5)\). The base of the fin is...
-
The post-closing trial balance for Cortez Co. is as follows. The subsidiary ledgers contain the following information: (1) accounts receivable'J. Anders $2,500, F. Cone $7,500, T. Dudley $5,000; (2)...
-
In today's workplace, it is important to develop diverse people skills. Managers and leaders need to manage diversity effectively. explain the following: In your opinion, what psychological process...
-
Skypes online platform for an easier experience for video conference users who do not need to setup and manage equipment, software, and networking systems exemplifies this type of value innovation:...
-
In a fixed effects experiment, suppose that there are n observations for each of four treatments. Let Q 2 1 , Q 2 2 , Q 2 3 be single-degree-of-freedom components for the orthogonal contrasts. Prove...
-
Refer to Problem 3-10. If we wish to detect a maximum difference in mean response times of 10 milliseconds with a probability of at least 0.90, what sample size should be used? How would you obtain a...
-
The Standard & Poor's 500 stock index is a commonly used measure of stock market performance in the United States. In the table below, we give the value of the S&P 500 index on the first day of...
-
The telecommunication sector is made up of companies that make communication possible on a global scale, whether it is through the phone or the Internet, through airwaves or cables, through wires or...
-
Identify a real (you may visit and use one of the listed cases) or fictitious case with an underpinning problem and address the following below: https://www.pmi.org/business-solutions/case-studies...
-
Given the data below ,what should the total salesforce be for my brand in thailand ? Should i go for total 9 or 147 ? Is it wise to share salesforce total evenly to all channels? Brand Indie Drug...
-
Molander Corporation is a distributor of a sun umbrella used at resort hotels. Data concerning the next month s budget appear below: Selling price per unit $ 2 4 Variable expense per unit $ 1 5 Fixed...
-
Michael is a senior student on job market. He is interested in a social media management position at Facebook. So before the interview, he decides to manage his own profile on Facebook and on...
-
Plate-glass insurance claims reveal a mean claim size of $120 and a standard deviation of $15. Assuming that the claim size X is normally distributed, calculate the size x of claim such that 800 of...
-
Determine which of the following limits exist. Compute the limits that exist. lim x-0 1- + 3x X
-
How many milliliters of 0.800 M KOH should be added to 5.02 g of 1,5-pentanedioic acid (C 5 H 8 O 4 , FM 132.11) to give a pH of 4.40 when diluted to 250 mL?
-
Calculate the pH of a 0.010 M solution of each amino acid in the form drawn here. H2N NH2 NH2 NH C=0 CH2 CH2 S CH, CH, CH2 CH2 (a) H;NCHCO, (b) H&NCHCO, (c) H,NCHCO, Glutamine Cysteine Arginine
-
(a) Draw the structure of the predominant form (principal species) of 1,3-dihydroxybenzene at pH 9.00 and at pH 11.00. (b) What is the second most prominent species at each pH? (c) Calculate the...
-
How do you prioritize features and attributes when designing a new product or service? What steps should be taken to ensure that the design process aligns with the organization's overall strategy and...
-
Information that does not have an adjustment reason code to route payments into the correct bank accounts to relay about patient benefit coverage to indicate the amount being paid and the date of...
-
Superior Company provided the following data for the year ended December 31 (all raw materials are used in production as direct materials): Selling expenses Purchases of raw materials Direct labor...

Study smarter with the SolutionInn App


