Using perfect induction, prove De Morgans theorem with four variables, specifically And WAX^y^2=wvxvjvz
Question:
Using perfect induction, prove De Morgan’s theorem with four variables, specifically
![]()
And
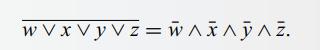
Transcribed Image Text:
WAX^y^2=wvxvjvz
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (7 reviews)
Omitting several uninteresting ca...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Digital Design Using VHDL A Systems Approach
ISBN: 9781107098862
1st Edition
Authors: William J. Dally, R. Curtis Harting, Tor M. Aamodt
Question Posted:
Students also viewed these Engineering questions
-
Use the Venn diagram to prove DeMorgans theorem, as given in expression 15b in Section 2.5. Section 2.5 In 1849 George Boole published a scheme for the algebraic description of processes involved in...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
The Crazy Eddie fraud may appear smaller and gentler than the massive billion-dollar frauds exposed in recent times, such as Bernie Madoffs Ponzi scheme, frauds in the subprime mortgage market, the...
-
Calculate the directional derivative in the direction of v at the given point. Remember to use a unit vector in your directional derivative computation. g(x, y, z) = xe-y, v = (1, 1, 1), P = (1,2,0)
-
Use the values of ÎHof in Appendix 4 to calculate ÎHo for the following reactions. a. b. Ca3(PO4)2(s) + 3H2SO4(l) 3CaSO4(s) + 2H3PO4(l) c. NH3(g) + HCl(g) NH4Cl(s) d. e. SiCl4(l) +...
-
Air is flowing in a wind tunnel at 15C, 80 kPa, and 200 m/s. The stagnation pressure at the probe inserted into the flow section is (a) 82 kPa (b) 91 kPa (c) 96 kPa (d) 101 kPa (e) 114 kPa
-
Evaluate the equilibrium constant at \(600 \mathrm{~K}\) for the reaction \[ \mathrm{CO}(\mathrm{g})+2 \mathrm{H}_{2}(\mathrm{~g}) ightarrow \mathrm{CH}_{3} \mathrm{OH}(\mathrm{g}) \] Data Table: 0...
-
Cassandra Corporation began operations on January 1, Year 1, as an online retailer of computer software and hardware. The following financial statement data were taken from Cassandras records at the...
-
Jack Sparrow, Will Turner, and James Norrington each want Davy Jones' heart for their own ends. The dead man's heart is hidden in a chest buried on Isla Cruces, but one needs the key to unlock the...
-
Draw a schematic for the following un simplified logic equation: f(x, y, z) = (x^y) V z.
-
Sketch the escape pattern of 32 wires (eight on each side) from a chip to a connector at a different part of a board. Assume all wires must be routed on the surface of the board and cannot cross over...
-
Use the test of your choice to determine whether the following series converge. 00 k=1 sin k
-
Hot tea (water) of mass 0.25 kg and temperature 73 C is contained in a glass of mass 200 g that is initially at the same temperature. You cool the tea by dropping in ice cubes out of the freezer that...
-
A 4.4810 3 kg train is stopped at a station. The train must go around a 181 m diameter corner right after it leaves the station. As the train leaves the station it provides a constant forward force...
-
In a Formula 1 race, a racer is driving her Honda F1 car (623 kg) out of a turn into a straight away. She accelerates from 16 m/s to 33 m/s over a distance of 113.0 m. There is an upward lift force...
-
An ideal gas contained in a piston cylinder undergoes two processes. The first process is a polytropic process with n = -1 which begins with p_1 = 2 bar, V_1 = 4 m^3 and ends at V_2 = 2 m^3. The...
-
A container with 2.9 kg of pure water at a temperature of 12 degrees Celsius is placed in a refrigerator where the air temperature is kept at 4 degrees Celsius. Using eBook Table 5.3, how much heat...
-
Wanda White acquired a sports equipment distribution business with a staff of six salespeople and two clerks. Because of the trust that Wanda had in her employees-after all, they were all her friends...
-
The MIT Sloan School of Management is one of the leading business schools in the U.S. The following table contains the tuition data for the masters program in the Sloan School of Management. a. Use...
-
Detection limit. In spectrophotometry, we measure the concentration of analyte by its absorbance of light. A low-concentration sample was prepared, and nine replicate measurements gave absorbances of...
-
An unknown sample of Ni 2+ gave a current of 2.36 A in an electrochemical analysis. When 0.500 mL of solution containing 0.028 7 M Ni 2+ was added to 25.0 mL of unknown, the current increased to 3.79...
-
An unknown sample of Ni 2+ gave a current of 2.36 A in an electrochemical analysis. When 0.500 mL of solution containing 0.028 7 M Ni 2+ was added to 25.0 mL of unknown, the current increased to 3.79...
-
Write down the bits that correspond to the network number (excluding the class prefix). Your answer should be only those bits, with no spaces. Enter your answer here
-
1. a. When interpreting individuals' scores on some measure why are standardized scores potentially beneficial compared to absolute raw scores? Why are z-scores also useful when computing and...
-
Lee has two main "math suites" in the SMC. One suite has offices for Professor Gladden and Professor Singletary. The other suite has offices for seven regular faculty. Proceeding in clockwise order...

Study smarter with the SolutionInn App


