Consider the equilibrium of a two-dimensional differential element in Cartesian coordinates, as shown in the following figure.
Question:
Consider the equilibrium of a two-dimensional differential element in Cartesian coordinates, as shown in the following figure. Explicitly sum the forces and moments and develop the two-dimensional equilibrium equations:
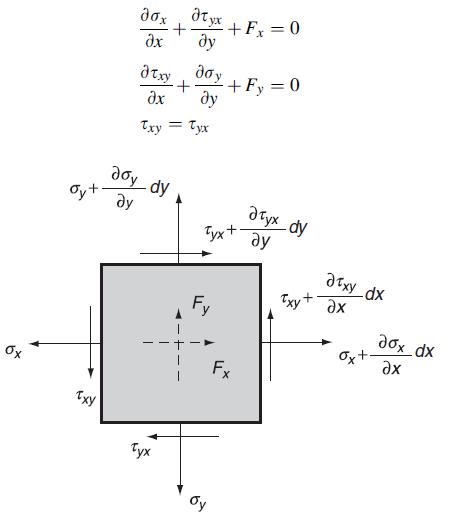
Transcribed Image Text:
Oyt Txy atyx ax + a txy + Txy = Tyx doy dy Tyx - + Fx = 0 Fy + +Fy = 0 Fx x_d Txy + Ox+ dox_dx
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
0 do o dxdyody X Ox atx T dydxdxFdxdy 0 dy Divide through by dxdy and let dxdy 0 ...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Elasticity Theory Applications And Numerics
ISBN: 9780128159873
4th Edition
Authors: Martin H. Sadd Ph.D.
Question Posted:
Students also viewed these Engineering questions
-
Following similar procedures as in Exercise 3.22 , sum the forces and moments on the two-dimensional differential element in polar coordinates (see figure), and explicitly develop the following...
-
Consider the two-dimensional case described in Exercise 3.22 with no body forces. Show that equilibrium equations are identically satisfied if the stresses are expressed in the form: where (x, y) is...
-
Consider the equilibrium of an oligomer: 2 MONOMERS DIMER The ÎH° for this reaction is positive and varies little between 4°C and 45°C. TÎS°, in contrast, varies widely...
-
In Problems 530, a. Classify the sequences as arithmetic, geometric, Fibonacci, or none of these. b. If arithmetic, give d; if geometric, give r; if Fibonacci, give the first two terms; and if none...
-
The tension in a wire clamped at both ends is doubled without appreciably changing the wire's length between the clamps. What is the ratio of the new to the old wave speed for transverse waves...
-
You will need the working papers that accompany this textbook in order to work this mini practice set. Bluma Co. uses a perpetual inventory system and both an accounts receivable and an accounts...
-
The piece of rubber is originally rectangular. Determine the average shear strain xy at A if the corners B and D are subjected to the displacements that cause the rubber to distort as shown by the...
-
Table shows a data set containing information for 25 of the shadow stocks tracked by the American Association of Individual Investors. Shadow stocks are common stocks of smaller companies that are...
-
Examine the role of Global Supply Chains and the related risks and opportunities in context to international business. Can residents live and work at the same place a tourist seeks enrichment,...
-
For a beam of circular cross-section, analysis from elementary strength of materials theory yields the following stresses: where R is the section radius, I = R 4 /4, M is the bending moment, V is the...
-
Starting with two-dimensional stress transformation relation (3.3.5), set dx/d = 0, and thus show that the relation to determine the angle to the principal stress direction p is given by tan 2 p = 2...
-
Things around you are emitting infrared radiation that includes the wavelength 9.9 x 10 -6 m. What is the energy of these IR photons?
-
An ideal monatomic gas is at \(125^{\circ} \mathrm{C}\). If you increase the thermal energy of the gas and the root-mean-square speed of the gas particles triples, by what factor does the absolute...
-
What is the root-mean-square speed of four monatomic ideal gas particles that have the following velocities (written in component form \():(4.0,6.0,2.0) \mathrm{m} / \mathrm{s},(8.0,-3.0,8.0)...
-
A theory predicts a reaction that produces ideal monatomic gas particles that have the following speeds: \(6.2 \mathrm{~m} / \mathrm{s}, 7. 4 \mathrm{~m} / \mathrm{s}, 7. 4 \mathrm{~m} / \mathrm{s},...
-
Initially, \(1.00 \mathrm{~mol}\) of an ideal monatomic gas has \(75.0 \mathrm{~J}\) of thermal energy. If this energy is increased by \(25.0 \mathrm{~J}\), what is the change in entropy?
-
A container holds \(1.0 \mathrm{~mol}\) of an ideal monatomic gas at \(72 \mathrm{~K}\), and then \(0.50 \mathrm{~mol}\) of another monatomic ideal gas at \(126 \mathrm{~K}\) is added. What is the...
-
John (age 51 and single) has earned income of $3,000. He has $30,000 of unearned (capital gain) income if he does participate in an employer-sponsored plan, what is the maximum deductible IRA...
-
Determine the annual percentage yield for a loan that charges a monthly interest rate of 1.5% and compounds the interest monthly.
-
How many milliliters of 0.800 M KOH should be added to 5.02 g of 1,5-pentanedioic acid (C 5 H 8 O 4 , FM 132.11) to give a pH of 4.40 when diluted to 250 mL?
-
Calculate the pH of a 0.010 M solution of each amino acid in the form drawn here. H2N NH2 NH2 NH C=0 CH2 CH2 S CH, CH, CH2 CH2 (a) H;NCHCO, (b) H&NCHCO, (c) H,NCHCO, Glutamine Cysteine Arginine
-
(a) Draw the structure of the predominant form (principal species) of 1,3-dihydroxybenzene at pH 9.00 and at pH 11.00. (b) What is the second most prominent species at each pH? (c) Calculate the...
-
6. Explain in detail why 8086 supports a maximum of 1MB physical memory? (2 mark) 7. What will be the capacity in megabytes of the physical memory of a microprocessor with a 28 bit address bus? What...
-
How does the use of dynamic process simulation differ from steady-state simulation, particularly in terms of modeling process transients and evaluating system robustness to disturbances ?
-
What role does process simulation play in the optimization of energy and material efficiency in chemical processes, and how can simulation tools help identify bottlenecks and opportunities for...

Study smarter with the SolutionInn App


