Following the steps outlined in the text, invert the form of Hookes law given by (4.2.7) and
Question:
Following the steps outlined in the text, invert the form of Hooke’s law given by (4.2.7) and develop form (4.2.10). Explicitly show that E = μ(3λ + 2μ)/(λ + μ) and v = λ/[2(λ + μ)].
Equation 4.2.7
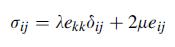
Equation 4.2.10
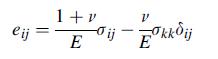
Transcribed Image Text:
gij = kekkoij +2u ij
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
ohe8 2le 322 427 120 e Ou 2 This ...View the full answer

Answered By

John Kimutai
I seek to use my competencies gained through on the job experience and skills learned in training to carry out tasks to the satisfaction of users. I have a keen interest in always delivering excellent work
4.70+
11+ Reviews
24+ Question Solved
Related Book For 

Elasticity Theory Applications And Numerics
ISBN: 9780128159873
4th Edition
Authors: Martin H. Sadd Ph.D.
Question Posted:
Students also viewed these Engineering questions
-
Using the results of Exercise 4.5, show that = E/ [2(1 + v)] and = Ev/ [(1 + v) (1 2v)]. Data from exercise 4.5 Following the steps outlined in the text, invert the form of Hookes law given by...
-
The Hampshire Company manufactures umbrellas that sell for $12.50 each. In 2014, the company made and sold 60,000 umbrellas. The company had fixed manufacturing costs of $216,000. It also had fixed...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
The fraud examiner's report should have what characteristics? a. Accuracy and clarity b. Relevant and material information c. Should not contain opinions or prejudices of fraud examiner. d. All of...
-
A uniform rope of mass m and length L hangs from a ceiling. (a) Show that the speed of a transverse wave on the rope is a function of y, the distance from the lower end, and is given by v = gy. (b)...
-
Presented below are two independent situations. Situation 1: Hatcher Cosmetics acquired 10% of the 200,000 ordinary shares of Ramirez Fashion at a total cost of $14 per share on March 18, 2019. On...
-
Should the requirements of the UCC be subject to the application of reliance theories? Go back and review the facts in Case 21-3 about the coal contract. Should silence followed by contract execution...
-
The investment committee of Sentry Insurance Co. is evaluating two projects, office expansion and upgrade to computer servers. The projects have different useful lives, but each requires an...
-
3. A 200 kg roller coaster starts from rest at the top of the first hill at a height of 20 m above the ground. The second hill is 15 m above the ground. a. A physics student in line for the ride...
-
For isotropic materials show that the principal axes of strain coincide with the principal axes of stress. Further, show that the principal stresses can be expressed in terms of the principal strains...
-
For isotropic materials, show that the fourth-order elasticity tensor can be expressed in the following forms: Cijkl = ijkl + (ldjk + dik jl) Cjk = ( + ji) + (k u) ijki (oiljk Cijkl E -ijk! + 2(1 +...
-
When the Canadian dollar depreciates, what happens to exports and imports in Canada?
-
Milton Industries expects free cash flows of $20 million each year. Miltons corporate tax rate is 35%, and its unlevered cost of capital is 9%. The firm also has outstanding debt of $104.13 million,...
-
Colt Systems will have EBIT this coming year of $18 million. It will also spend $7 million on total capital expenditures and increases in net working capital, and have $4 million in depreciation...
-
Consider a simple firm that has the following market-value balance sheet: Next year, there are two possible values for its assets, each equally likely: $1220 and $970. Its debt will be due with 5.1%...
-
Empire Industries forecasts net income this coming year as shown below (in thousands of dollars): Approximately $150,000 of Empires earnings will be needed to make new, positive- NPV investments....
-
NatNah, a builder of acoustic accessories, has no debt and an equity cost of capital of 17%. NatNah decides to increase its leverage to maintain a market debt-to-value ratio of 0.4. Suppose its debt...
-
How may beta coefficients be used to standardize returns for risk to permit comparisons of mutual fund performance?
-
H Corporation has a bond outstanding. It has a coupon rate of 8 percent and a $1000 par value. The bond has 6 years left to maturity but could be called after three years for $1000 plus a call...
-
Using activities, find the concentrations of the major species in 0.10 M NaClO 4 saturated with Mn(OH) 2 . Take the ionic strength to be 0.10 M and suppose that the ion size of MnOH + is the same as...
-
Explain why the solubility of an ionic compound increases as the ionic strength of the solution increases (at least up to ~ 0.5 M).
-
Which statements are true? In the ionic strength range 00.1 M, activity coefficients decrease with (a) Increasing ionic strength; (b) Increasing ionic charge; (c) Decreasing hydrated radius.
-
The XYZ Corporation, a manufacturing company, uses accelerated depreciation for tax purposes but straight-line depreciation for financial reporting. As a result, the company's income tax expense...
-
Use the diagram to find the following bearings. 112 O 47 B 28 C
-
On December 2, John Leahy received his bank statement showing a balance of $358.97. His checkbook showed a balance of $479.39. There was a check printing charge of $13.95, and interest earned was...

Study smarter with the SolutionInn App


