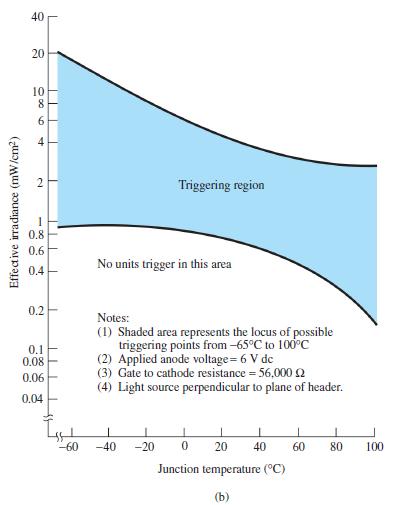
a. Using Fig. 17.24b, determine the minimum irradiance required to fire the device at room temperature (25C).
Question:
a. Using Fig. 17.24b, determine the minimum irradiance required to fire the device at room temperature (25°C).
b. What percentage reduction in irradiance is allowable if the junction temperature is increased from 0°C (32°F) to 100°C (212°F)?
Fig. 17.24b
Transcribed Image Text:
40 20 10 Triggering region 0.8 0.6 No units trigger in this area 0.4 0.2 Notes: (1) Shaded area represents the locus of possible triggering points from -65°C to 100°C (2) Applied anode voltage= 6 V de (3) Gate to cathode resistance = 56,000 (4) Light source perpendicular to plane of header. 0.1 0.08 0.06 0.04 -60 -40 -20 20 40 60 80 100 Junction temperature (°C) (b) TIL | 2. Effective irradiance (mW/cm2)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (9 reviews)
a Using Fig 1724b determine the minimum irradiance required to fire the device at room temper...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Electronic Devices And Circuit Theory
ISBN: 9781292025636
11th Edition
Authors: Robert Boylestad, Louis Nashelsky
Question Posted:
Students also viewed these Engineering questions
-
A room is 25 feet by 32 feet. How much will it cost to cover the floor with carpet costing $12 a square yard (9 square feet), if 4 extra square yards are needed for matching? If a portion of a...
-
If you place water at room temperature in a well-insulated cup and allow some of the water to evaporate, the temperature of the water in the cup will drop lower than room temperature. Come up with an...
-
What temperature is required to obtain 0.50% C at a distance of 0.5 mm beneath the surface of a 0.20% C steel in 2 h, when 1.10% C is present at the surface? Assume that the iron is FCC?
-
Why do the requirements drift once a project is under way?
-
What is the user cost of capital?
-
Develop three cost classifications for the LCC of an office building.
-
Estimate the power that the human heart must impart to the blood to pump it through the two carotid arteries from the heart to the brain. List all assumptions and show all calculations.
-
Morris Supply Company had the following transactions in 2010. 1. Acquired $50,000 cash from the issue of common stock. 2. Purchased $210,000 of merchandise for cash in 2010. 3. Sold merchandise that...
-
A physics student stands on a cliff overlooking a lake and decides to throw a golf ball to her friends in the water below. She throws the golf ball with a velocity of 22.5m/s at an angle of 32.5...
-
Subject : Strategic Management in a Global Environment Safaricom: Innovative Telecom Solutions to Empower Kenyans As the largest mobile provider in Kenya, Safaricom has touched the lives of Kenyans...
-
For the network of Fig. 17.28, if V BR = 6 V, V = 40 V, R = 10 k, C = 0.2 mF, and V GK (firing potential) = 3 V, determine the time period between energizing the network and the turning on of the S...
-
a. In Fig. 17.22, if V Z = 50 V, determine the maximum possible value the capacitor C 1 can charge to (VGK 0.7 V). b. Determine the approximate discharge time (5t) for R 3 = 20 k. c. Determine the...
-
How much would Barry (from Problem 1) have if he could earn a 10 percent return, compounded monthly, on his investment beginning at age 35? In problem (1) Barry has just become eligible for his...
-
An investor buys 200,000 shares of a private placement that's sold under Regulation D. The investor is not an officer or director of the company. How many shares is the investor permitted to sell...
-
Step 1: Self-Assessment Begin by taking an objective assessment of your current nonverbal skills. What do you feel are your strengths? What do you feel are areas for improvement? Write up your...
-
Think of a time that you witnessed emotional contagion, either in person or online. Explain this experience detail, discuss what caused it, and discuss the impact of this emotional contagion (whether...
-
Early AI developers measured the intelligence of the technology by its ability to play games, such as chess. Why might this definition of intelligence be limiting? What other forms of intelligence...
-
These rituals reinforce the important feeling of belonging to out most basic group, our family. What is one of your family's rituals that you really enjoy?
-
What changes in accounting method can be made without IRS approval?
-
a. Determine the domain and range of the following functions.b. Graph each function using a graphing utility. Be sure to experiment with the window and orientation to give the best perspective of the...
-
(a) Write a balanced equation for the reaction and calculate E for the reaction. (b) Predict whether an equimolar mixture of PuO 2 2 + and PuO + 2 will oxidize H 2 O to O 2 at a pH of 2.00 and P O2 =...
-
Calculate the voltage of the following cell, in which KHP is potassium hydrogen phthalate, the monopotassium salt of phthalic acid. By the reasoning in Figure 13-8, in which direction do electrons...
-
The following cell has a voltage of 0.083 V: Hg(l) | Hg(NO3)2(0.001 0 M), KI(0.500 M) || S.H.E. From this voltage, calculate the equilibrium constant for the reaction In 0.5 M KI, virtually all the...
-
Alice is single and self-employed in 2023. Her net business profit on her Schedule C for the year is $192,800. What are her self-employment tax liability and additional Medicare tax liability for...
-
D.L. Ko and Company, a manufacturer of quality handmade walnut bowls, has had a steady growth in sales for the past 5 years. However, increased competition has led Mr. Ko, the president, to believe...
-
McCullough Hospital uses a job-order costing system to assign costs to its patients. Its direct materials include a variety of items such as pharmaceutical drugs, heart valves, artificial hips, and...

Study smarter with the SolutionInn App


