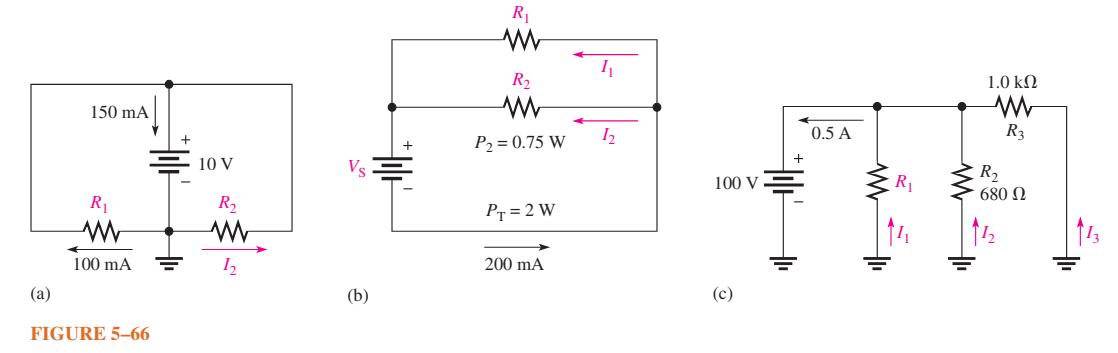
Find the values of the unspecified quantities (shown in color) in each circuit of Figure 566. (a)
Question:
Find the values of the unspecified quantities (shown in color) in each circuit of Figure 5–66.
Transcribed Image Text:
(a) 150 mA R₁ 100 mA FIGURE 5-66 = 10 V R₂ www 12 (b) R₁ R₂ www P₂ = 0.75 W PT = 2 W 200 mA 1₁ 1₂ 100 V (c) 0.5 A R₁ 1.0 ΚΩ www R3 R₂ 680 Ω
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)
Lets analyze each circuit in Figure 566 to find the values of the unspecified quantities shown in color Well apply the principles of Ohms law and the ...View the full answer

Answered By

Deepak Sharma
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
In the circuit in Fig. 3.102, find the values of R, V1, and V2 given that io = 18 mA. Figure 3.102 100 V
-
1. ABM's breakeven point is 4,000 units at a sales price of P50 per unit, variable cost of P30 per unit, and total fixed costs of P80,000. If ABM sells 500 additional units, by how much will profit...
-
In Figure particles are shown traveling counterclockwise in circles of radius 5 m. The acceleration vectors are indicated at three specific times. Find the values of v and dv/dt for each of these...
-
Calculate the heat required to raise 50 kg of solid sodium carbonate (Na2CO3) from 10C to 50C at 1atm using (a) The true heat capacity of Na2CO3, which is 1.14 kJ / (kg C). (b) A heat capacity...
-
Each of the following independent events requires a year-end adjusting entry. Record each event and the related adjusting entry in general journal format. The first event is recorded as an example....
-
The manager of a branch office of Polar Bank observed that 20 customers per hour, on average, arrive during peak time, and that there are about four customers, on average, in the bank branch at any...
-
For each of the experiments described below, identify any problems and explain how the problems could have been avoided. a. A new drug for attention deficit disorder (ADD) is supposed to make...
-
The balance sheet of Sco appeared as follows on March 1, 2011, when an interim trustee was appointed by the U.S. trustee to assume control of Sco's estate in a Chapter 7 case. ADDITIONAL...
-
Instruction: Refer to any of the marketing plan templates given. Prepare a complete marketing plan for new product launching. Tip: A good Marketing Plan includes these 10 elements: 1. Describe Your...
-
In the circuit of Figure 564, determine resistances R 2 , R 3 , and R 4 . Data in Figure 564 +0 Vs 7.5 mA 1 mA FIGURE 5-64 2 mA R 50 0.5 mA R www R3 R4
-
What is the total resistance between terminal A and ground in Figure 567 for the following conditions? (a) SW1 and SW2 open (b) SW1 closed, SW2 open (c) SW1 open, SW2 closed (d) SW1 and SW2 closed. ...
-
Use a CAS to find sin 5 x cos 2 x dx.
-
Why might there be two different component costs for common equity? Which one is generally relevant, and for what type of firm is the second one likely to be relevant?
-
Would you expect an abandonment option to increase or decrease a projects expected NPV and risk (as measured by the coefficient of variation)? Explain.
-
Like the boards of most corporations, the board of directors of Paolos Pizzas, Inc. conducts business through a. annual shareholders meetings. b. consultations with corporate officers and employees....
-
If you were analyzing a replacement project and you suddenly learned that the old equipment could be sold for $1,000 rather than $100, would this new information make the replacement look better or...
-
In what ways is the setup for finding a projects cash flows similar to the projected income statements for a new single-product firm? In what ways would the two statements be different?
-
The thallium (present as Tl 2 SO 4 ) in a 9.486-g pesticide sample was precipitated as thallium(I) iodide. Calculate the mass percent of Tl 2 SO 4 in the sample if 0.1824 g of TlI was recovered.
-
What is the maximum volume of 0.25 M sodium hypochlorite solution (NaOCl, laundry bleach) that can be prepared by dilution of 1.00 L of 0.80 M NaOCl?
-
Edit the structure array created in Problem 48 to change the maintenance data for the Clark St. bridge from 2012 to 2018. Problem 48 Create a structure array that contains the following information...
-
Add the following bridge to the structure array created in Problem 48. Problem 48 Create a structure array that contains the following information fields concerning the road bridges in a town: bridge...
-
Suppose that y = -3 + ix. For x = 0, 1, and 2, use MATLAB to compute the following expressions. Hand-check the answers. a. |y| b. y c. (-5 7i)y d. y/6 3i
-
Research on childrens advertising literacy needs to keep pace with the emergence of different forms of marketing communication in the digital era. This paper reports on a qualitative study conducted...
-
Many different leadership models stress the importance of the leader being an outgoing (or extroverted) individual. Explain why is this is considered essential to effective leadership?
-
Using a generic project management 3-Dimensional maze, critically identify 5 forces (at least 1 from each layer) which were faced by the DIA project and how you could have dealt with them.

Study smarter with the SolutionInn App


