Use a commercial flowchart simulation program such as HYSYS or ASPEN to simulate the ammonium nitrate manufacturing
Question:
Use a commercial flowchart simulation program such as HYSYS or ASPEN to simulate the ammonium nitrate manufacturing process described in Example 10.3-3.
Example 10.3-3
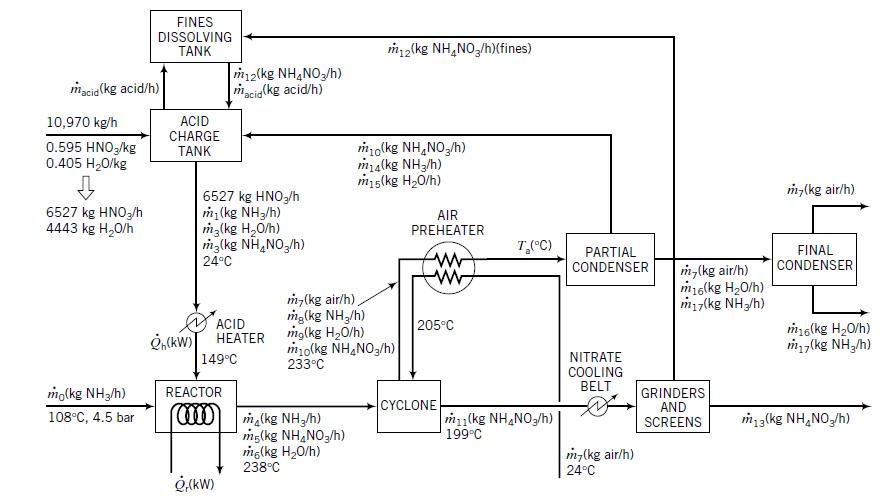
Transcribed Image Text:
FINES DISSOLVING TANK m2(kg NH,NO,/h)(fines) m12(kg NH,NO,/h) macig(kg acid/h) macia (kg acid/h) 10,970 kg/h ACID CHARGE 0.595 HNO,/kg 0.405 H20/kg mo(kg NH,NO,/h) ma(kg NH3/h) m15(kg H20/h) TANK m,(kg air/h) 6527 kg HNO3/h 4443 kg H20/h 6527 kg HNO,/h m (kg NH3/h) m3(kg H,0/h) m3(kg NH,NO,/h) AIR PREHEATER T,(C) PARTIAL CONDENSER FINAL CONDENSER 24°C m,(kg air/h) m16(kg H20/h) miy(kg NH/h) m,(kg air/h), mg(kg NH,/h) mg(kg H20/h) m10(kg NH,NO3/h) 233°C ACID НЕАTER 205°C m6(kg H20/h) m (kg NH,/h) NITRATE COOLING BELT 149°C mo(kg NH3/h) REACTOR GRINDERS AND SCREENS CYCLONE m1(kg NH,NO3/h)| 199°C 108°C, 4.5 bar ma(kg NH,/h) mg(kg NH,NO,/h) mg(kg H20/h) 238°C m3(kg NH,NO,/h) m-(kg air/h) 24°C
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Elementary Principles of Chemical Processes
ISBN: 978-0471720638
3rd Edition
Authors: Richard M. Felder, Ronald W. Rousseau
Question Posted:
Students also viewed these Business questions
-
Repeat Exercise 5 assuming that the size of each batch is 100 instead of 10,000. Compare the control chart to the one found for Exercise 5. Comment on the general quality of the manufacturing process...
-
Ammonium nitrate, NH4NO3, is used as a nitrogen fertilizer and in explosives. What is the molar mass of NH4NO3?
-
The throttle process described in Example 6.5 is an irreversible process. Find the entropy generation per kg ammonia in the throttling process.
-
What do you think people would say about Corrie from the few quotes we have from her book? What was her personality like? Do you think she handled her incarceration differently than Elie Wiesel?...
-
(a) At 800 K the equilibrium constant for I2(g) 2 I(g) is Kc = 3.1 Ã 10-5. If an equilibrium mixture in a 10.0-L vessel contains 2.67 Ã 10-2 g of I(g), how many grams of I2 are in the...
-
2. Your firm processes 50,000 checks each year with an average face.value of $50 per check. Col- lecting on these checks requires two days of mail float, three days of processing float, and one day...
-
A New York firm is offering a new financial instrument called a "happy call." It has a payoff function at time \(T\) equal to \(\max (.5 S, S-K)\), where \(S\) is the price of a stock and \(K\) is a...
-
A partial adjusted trial balance of Frangesch Company at January 31, 2017, shows the following. Instructions Answer the following questions, assuming the year begins January 1. a) If the amount in...
-
Image transcription text KSBN 101754Z 32011KT 10SM BKN018 BKN022 OVC036 17/12 A2997 RMK A02 SLP147 TO1670122 10167 20133 50012 $ BKN018 BKN022 0VC036 10167 20133 50012 $ KMDW 101753Z VRBO4KT 10SM...
-
The manager of the BiLo Supermarket in Mt. Pleasant, Rhode Island, gathered the following information on the number of times a customer visits the store during a month. The responses of 51 customers...
-
You are to write the code for a convergence module that can deal with one to three tear stream variables using theWegstein algorithm, as outlined in Appendix A.2. The object is to determine the...
-
On an oversized page, draw a flowchart for this process. Label each stream with an identifying symbol (e.g., S l , S 2 , S 3 , ...) and known information about what the stream is and/or what it...
-
For the following exercises, find f 1 (x) for each function. f(x) = x/x + 2
-
At 922 K, the enthalpy of liquid Mg is 26.780 [kJ/mol] and the entropy is 73.888 [J/(mol K)]. Determine the Gibbs energy of liquid Mg at 1300 K. The heat capacity of the liquid is constant over this...
-
What are the important changes that have occurred in stock markets since 2005?
-
The Nelson partnership begins business on July 17, 2018. It is not able to use business purpose to support a tax year. The four partners, their interests in partnership capital and profits, and their...
-
Baker, a cash-basis, calendar-year taxpayer, is a partner in an accrual-basis partnership that reports its taxable income on an October 31 fiscal year. Baker has been provided the following...
-
A binary mixture of carbon dioxide and water exists in vaporliquid equilibrium at 343.15 K and 1 bar. The solubility of CO2 in the liquid has been measured as xCO2 = 0.000255. What is the Henrys law...
-
The following unadjusted trial balance is prepared at fiscal year-end for Foster Products Company. Rent expense and salaries expense are equally divided between selling activities and general and...
-
Consider the combustion of methanol below. If 64 grams of methanol reacts with 160 grams of oxygen, what is the CHANGE in volume at STP. 2CH3OH(g) + 3O2(g) 2CO2(g) + 4H2O(1) The volume decreases by...
-
If the current in an electric conductor is 2.4 A, how many coulombs of charge pass any point in a 30 second interval?
-
Determine the time interval required for a 12-A battery charger to deliver 4800 C.
-
A lightning bolt carrying 30,000 A lasts for 50 microseconds. If the lightning strikes an airplane flying at 20,000 feet, what is the charge deposited on the plane?
-
Linux - choose how you will manage your software install packages? Analyse some advantages of using a package manager to manage software?
-
A firm produces output according to the production function Q = K1/2 1/2. The demand schedule for the output is P= 20 - Q. If K is fixed at 4 units, what is this firm's short-run demand curve for...
-
Write about the Biden administration's State of the Union address by taking one side and examining how it affects you as an individual. Discuss the policy, economic accomplishments, what he has done...

Study smarter with the SolutionInn App



